Thalidomide is a chiral molecule, which was sold in the 1960s as a sedative in its (S,R)-racemic form. The tragedy was that the (S)-isomer was tetragenic, and only the (R) enantiomer acts as a sedative. What was not appreciated at the time is that interconversion of the (S)- and (R) forms takes place quite quickly in aqueous media. Nowadays, quantum modelling can provide good in-silico estimates of the (free) energy barriers for such processes, which in this case is a simple keto-enol tautomerism. In a recently published article[1], just such a simulation is reported. By involving two explicit water molecules in the transition state, an (~enthalpic) barrier of 27.7 kcal/mol was obtained. The simulation was conducted just with two water molecules acting as solvent, and without any additional continuum solvation applied. So I thought I would re-evaluate this result by computing it at the ωB97XD/6-311G(d,p)/SCRF=water level (a triple-ζ basis set rather than the double-ζ used before[1]), and employing a dispersion-corrected DFT method rather than B3LYP.
Keto-enol tautomerisation occupies a unique position in the history of mechanistic chemistry[2]. In 1889, Beckmann got the whole field rolling by proposing an inferred enol intermediate (which he did not observe) to explain the isomerism of menthone to iso-menthone in conc. sulfuric acid. In modelling the enolisation of thalidomide, I have used both implicit and explicit solvents acting in a self-consistent manner. This approach is not yet much adopted in the wider literature[3]. I have deployed it extensively in this blog as an encouragement to others (selected examples are listed at the bottom of this post). It is worth noting at the outset that the transition state reported previously[1] has a computed dipole moment of ~10D. My experience[3] suggests that any geometry with a dipole moment of this magnitude (or greater) is likely to relax when placed into a continuum field, and this relaxation becomes an important perturbation of both the computed geometry of the transition state and the intrinsic reaction coordinate profile computed from that starting point.
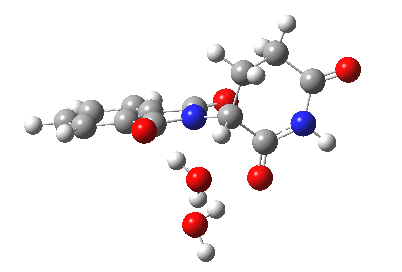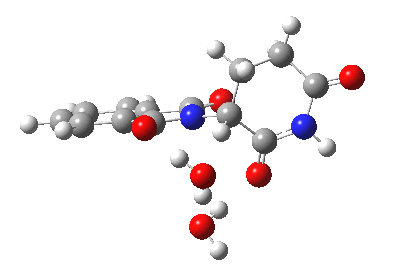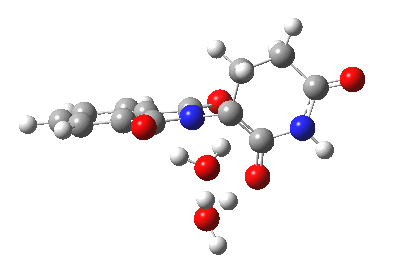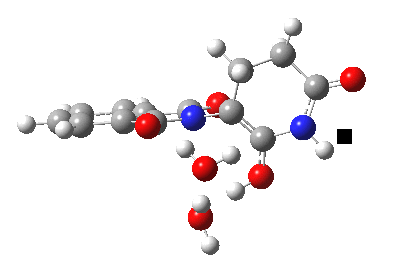
The (re)computed geometry of the aqueous transition state for enolisation of thalidomide is shown below, and for which the entropy-corrected ΔG†298 is 31.0 kcal/mol (the barrier for the prototypical enolisation of propanone is computed as 34.4 kcal/mol). The value in the literature[1] is given as 27.7 kcal/mol for the zero-point-energy corrected total energy barrier, but this value notably does NOT include any entropic corrections. The measured literature value for ΔG†298 is reported as 24.3 kcal/mol at pH 8, a value which probably also includes contributions from both the pure water catalysed route and those from hydroxide anion catalysis (see below). At this point, I should remind that the free energy of activation for a bi- or termolecular reaction in solution must be obtained by correcting the value obtained for a standard state of 1 atmosphere (the state used for the value quoted above). According to Alvarez-Idaboy and co-workers[4], this amounts to a total correction of -4.5 kcal/mol for a bimolecular reaction, and -8.73 kcal/mol for a termolecular reaction. Where one of the components of a termolecular reaction is also the solvent, these corrections probably need to be themselves reduced. But this does achieve a reduction in the computed value of 31.0 kcal/mol to something quite close to the experimental value!
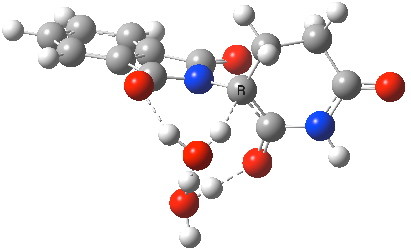
Aqueous transition state for enolisation of thalidomide. Click for 3D.
Next, I want to consider the base-catalysed enolisation pathway. As with the reaction of dichlorobuteneone with tolyl-thiolate about which I wrote in another post, the authors of the thalidomide study[1] modelled this route by introducing a solvated hydroxide anion, “OH–·H2O” into the structure without any accompanying counter-ion. In other words, their total system has an overall negative charge. I argued before, and I argue again here, that there is no real need to have to do this. Why not for example introduce NaOH•H2O instead? One might argue that the cationic counter-ion so introduced cannot be properly modelled, but the combination of explicit first-sphere water molecules coupled with a continuum model actually handles these counter-ions reasonably well. So may I introduce you to my version of the base-catalysed reaction, involving a contact-ion-pair:
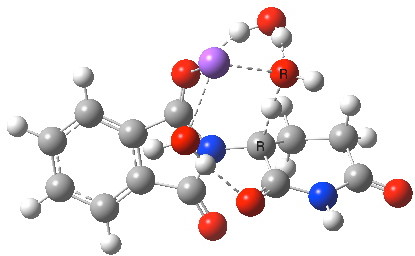
Base-catalysed (NaOH) enolisation of thalidomide. Click for 3D.
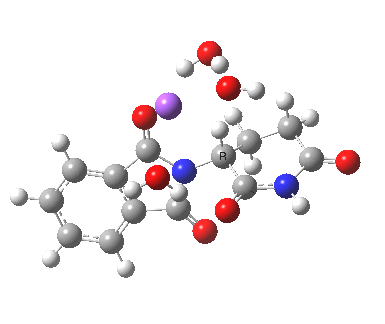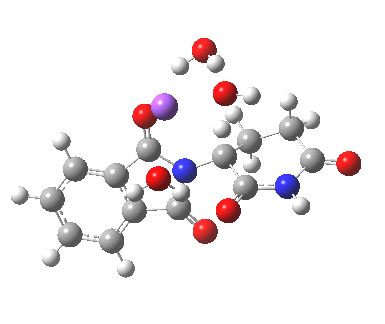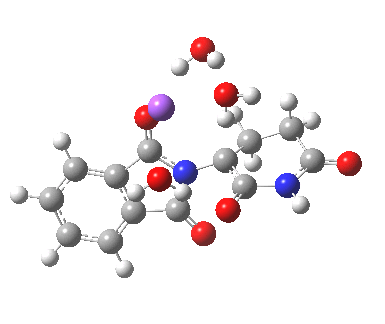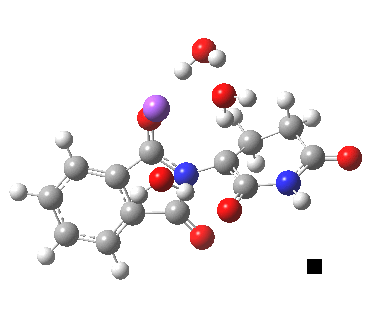
This has ΔG†298 4.7 kcal/mol, much lower than the neutral water catalysed reaction. This value is of course for a standard state for [Na+OH–] (1 atm). At pH 8, [OH–] is at least six orders of magnitude less, which may rationalise why the experimental rate is so much slower than this barrier might imply. The IRC corresponds to proton transfer.
I would like to end by noting that many mechanisms which would otherwise involve the development of charge-separation may well borrow a protic solvent molecule in the manner shown here to reduce the degree of charge-separation needed. Further examples of this are listed below.
- Oxime formation from hydroxylamine and ketone. Part 2: Elimination.
- Oxime formation from hydroxylamine and ketone: a (computational) reality check on stage one of the mechanism.
- Transition state models for Baldwin dig(onal) ring closures.
- Transition state models for Baldwin’s rules of ring closure.
- The mechanism (in 4D) of the reaction between thionyl chloride and a carboxylic acid.
- Mechanism of the diazomethane alkylation of a carboxylic acid.
- The mechanism of the Baeyer-Villiger rearrangement.
- Stereoselectivities of Proline-Catalyzed Asymmetric Intermolecular Aldol Reactions.
- Secrets of a university tutor: tetrahedral intermediates.
References
- C. Tian, P. Xiu, Y. Meng, W. Zhao, Z. Wang, and R. Zhou, "Enantiomerization Mechanism of Thalidomide and the Role of Water and Hydroxide Ions", Chemistry – A European Journal, vol. 18, pp. 14305-14313, 2012. https://doi.org/10.1002/chem.201202651
- E. Beckmann, "Untersuchungen in der Campherreihe", Justus Liebigs Annalen der Chemie, vol. 250, pp. 322-375, 1889. https://doi.org/10.1002/jlac.18892500306
- J. Kong, P.V.R. Schleyer, and H.S. Rzepa, "Successful Computational Modeling of Isobornyl Chloride Ion-Pair Mechanisms", The Journal of Organic Chemistry, vol. 75, pp. 5164-5169, 2010. https://doi.org/10.1021/jo100920e
- J.R. Alvarez-Idaboy, L. Reyes, and J. Cruz, "A New Specific Mechanism for the Acid Catalysis of the Addition Step in the Baeyer−Villiger Rearrangement", Organic Letters, vol. 8, pp. 1763-1765, 2006. https://doi.org/10.1021/ol060261z
Tags: 298 4.7, aqueous media, ATM, energy barrier, energy barriers, free energy, Historical, Reaction Mechanism, simulation, zero-point-energy
There is “a typo” in the drawing of the phthalimido group for both enantiomers of thalidomide.