These four posts (the box set) set out to try to define the energetics for a reasonable reaction path for the Willgerodt-Kindler reaction. The rate of this reaction corresponds approximately to a free energy barrier of ~30 kcal/mol. Any pathway found to be >10 kcal/mol at its highest point above this barrier was deemed less probable. The first three efforts at defining such pathways all gave such a result. Here I try a fourth pathway in search of the hitherto elusive appropriately low energy barrier.
The previously explored pathway invoked an aziridinium cation as an intermediate (Int4). The challenge now is to define a route into this intermediate and another out of it to lead to the eventual product. I previously explored the energy of forming Int4 by first using the lone pair of electrons on the nitrogen to form the ring, resulting in a carbanionic ylid which only then gained a proton to form Int4. This time I will try reversing this sequence, by protonation first to form Int5 via TS4, forming a cation resonance stabilized via sulfur. Only then does the nitrogen lone pair come into play to form the ring via TS8. The free energy barriers for both these species are now within a reasonable range, being within 10 kcal/mol of the estimated rate barrier (FAIR data for this pathway collected at DOI: 10.14469/hpc/7385). It is also important to note that this is only an exploratory model, which has not yet been “optimized”. Thus to reduce the computer time needed, ammonia is used as a model base. The full model would use morpholine, which as a better base might be expected to eg reduce the barrier for TS4. Also, these are bimolecular reactions computed for a standard state of ~0.04M. More concentrated solutions would also reduce the barrier. The anion present along the entire reaction pathway is not included in this model; doing so might also alter slightly the barriers.
Having found a reasonable route to Int4, it now has to be converted in the first instance to Int6, which is then easily protonated to the initial product, leading eventually to the thioamide outcome of this reaction. After much exploration, a good route was found to unexpectedly involve Int7. This is formed by ring opening of Int4 via TS9, with the sulfur migrating along the carbon chain in preference to forming the rather less resonance stabilized benzylic cation. Int7 then reverses this migration, with the base removing a proton and the sulfur migrating back to the carbon atom it had started from in Int4 via TS10. Both these barriers are also <10 kcal/mol of the barrier inferred from the reaction rate.
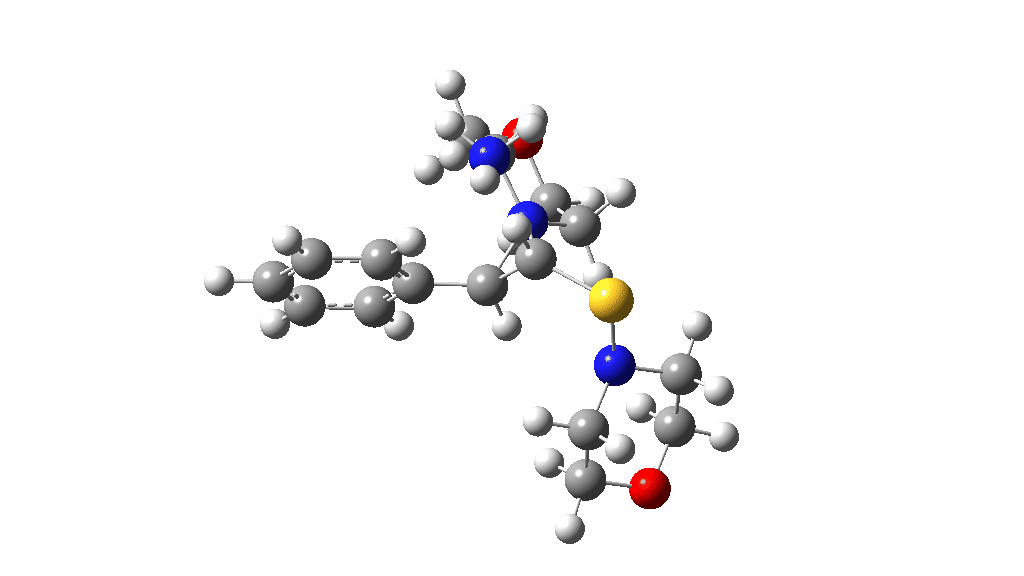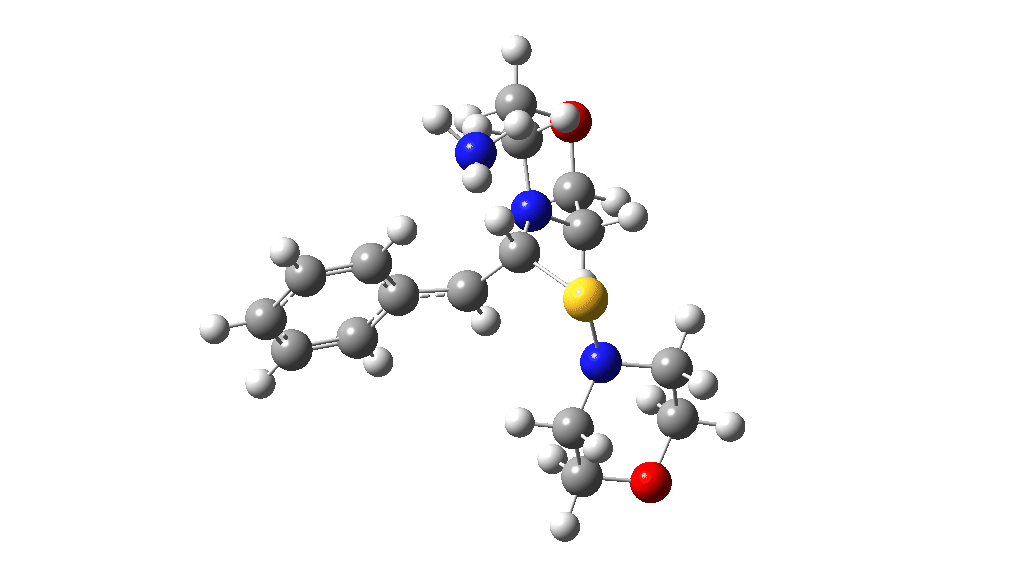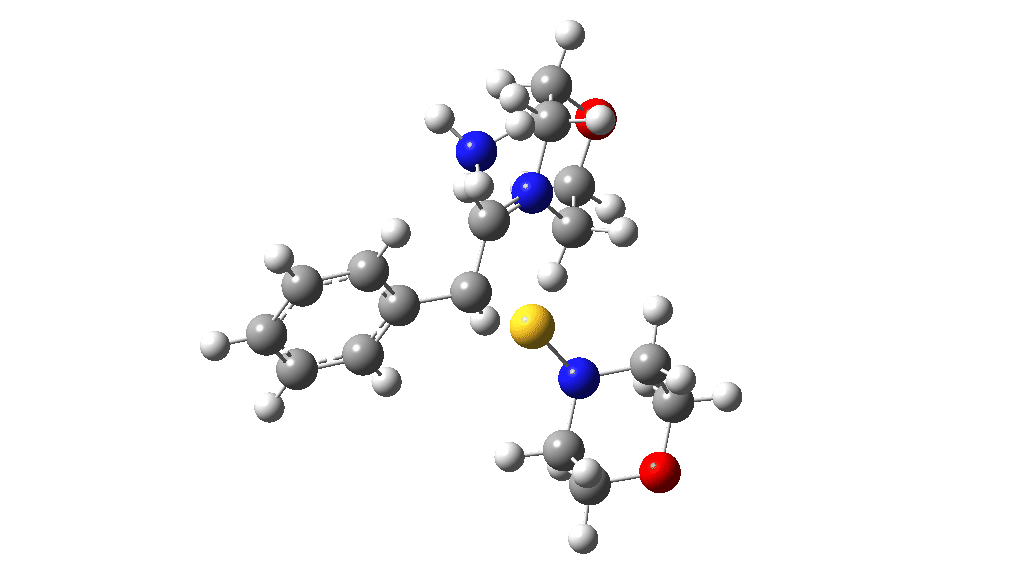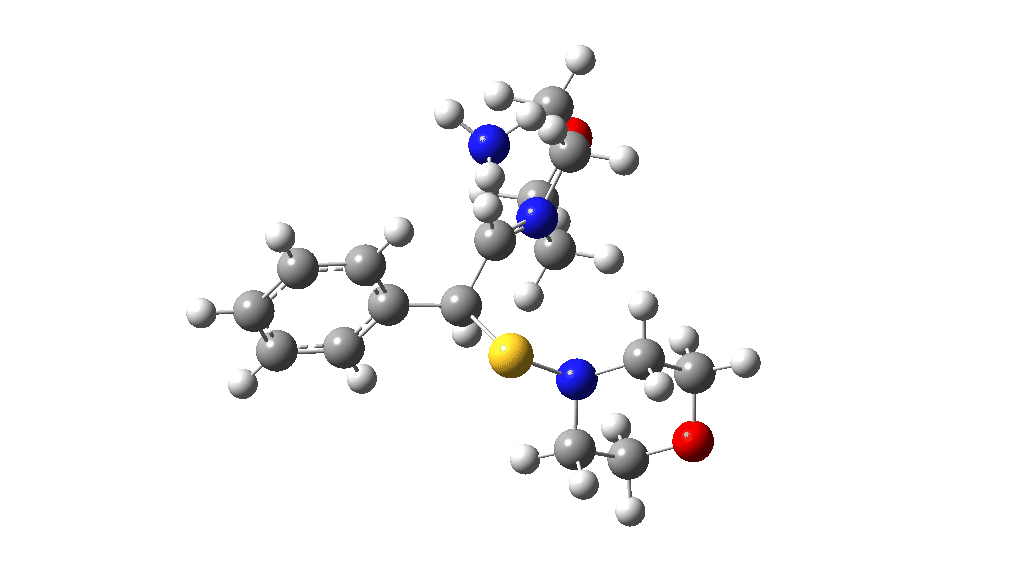
TS9
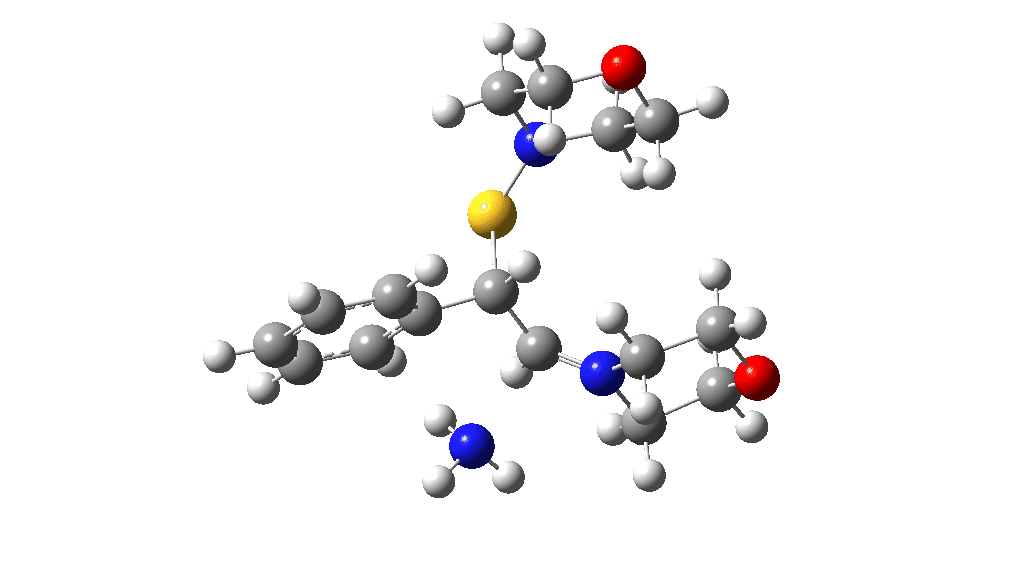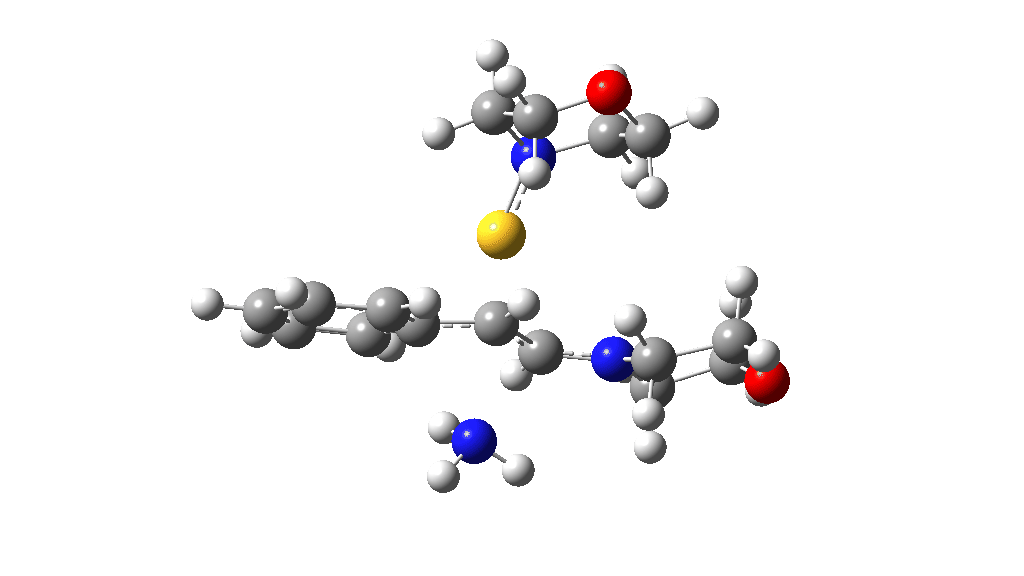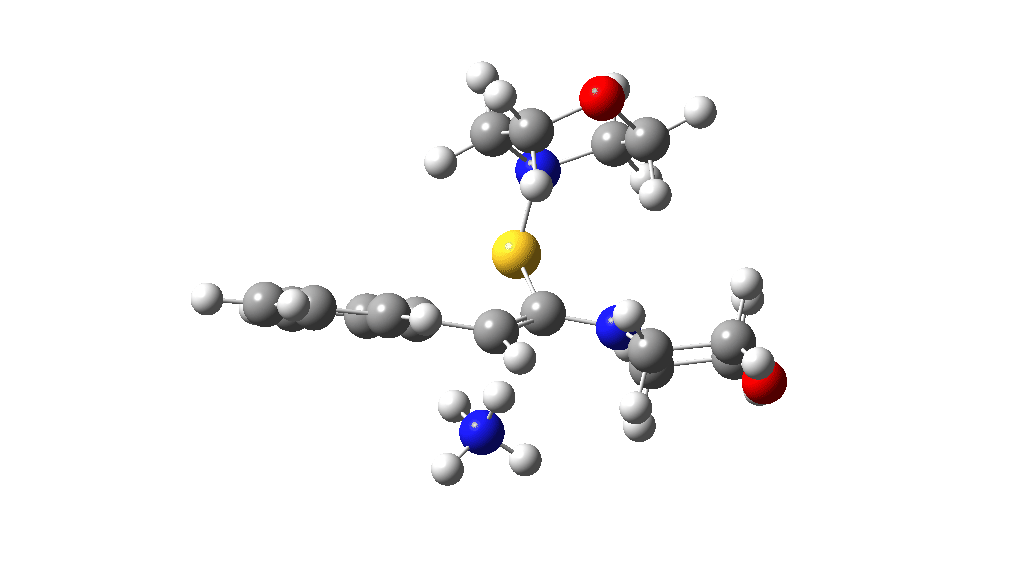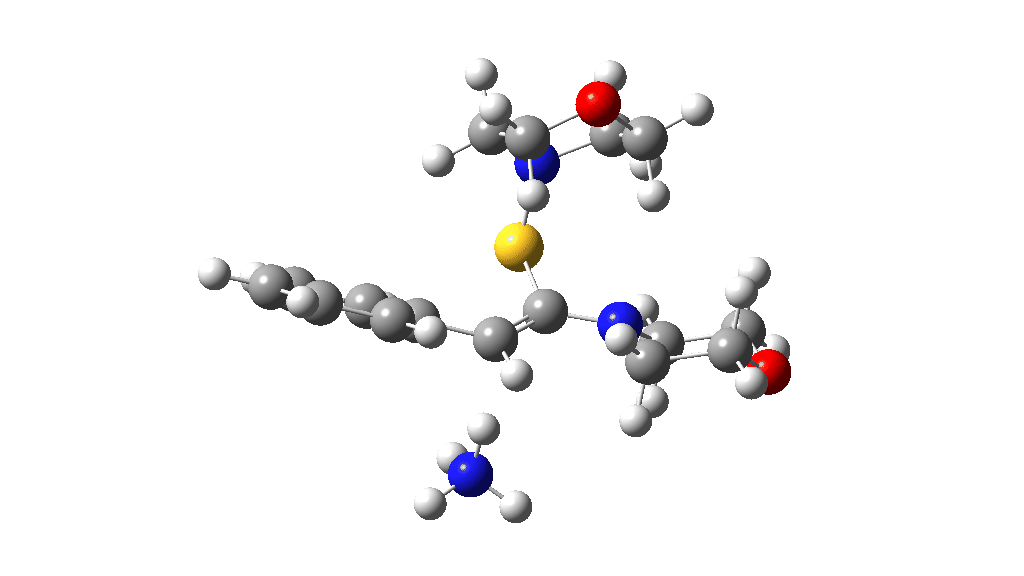
TS10
We thus finally have a model which is in accord with the kinetics of this reaction. As noted above, the model can always be refined further by eg improving the base, searching for lower energy conformers of the various transition states etc. Such optimisation can often reduce barriers further by perhaps 3-4 kcal/mol, along with the aforementioned reduction of a bimolecular reaction by increasing concentrations.
I hope this “box set” of mechanistic investigations gives some insight into how a reaction can be explored using calculations. In this instance we also have the benefit that our final mechanism does lead to an interesting prediction. Thus Int7 is predicted to be almost as stable as the final product. Perhaps its presence might be detectable if searched for. Identifying some Int7 in the reaction products would certainly provide good supporting evidence for our conclusions.
The DOI for this post is 10.14469/hpc/7387
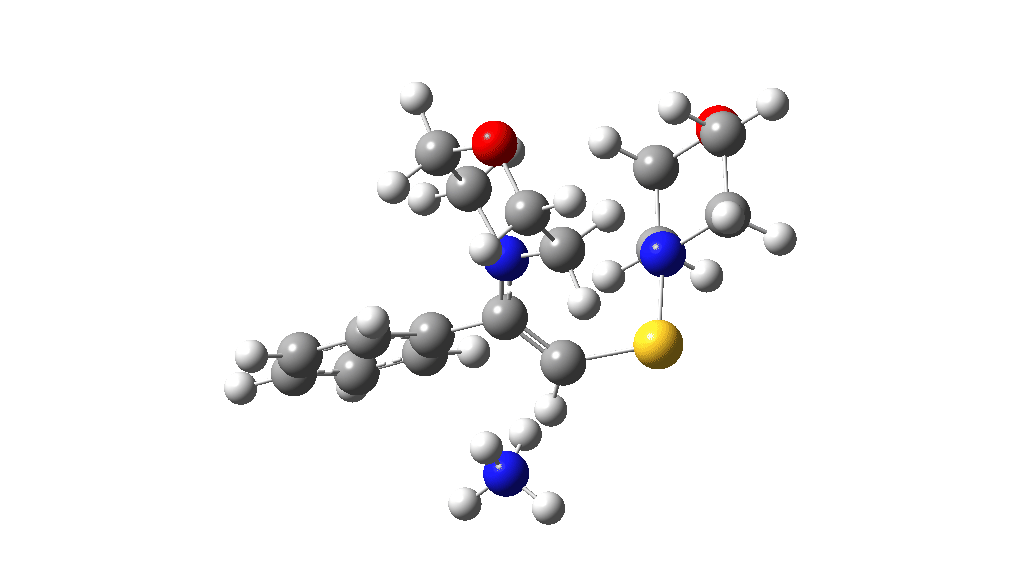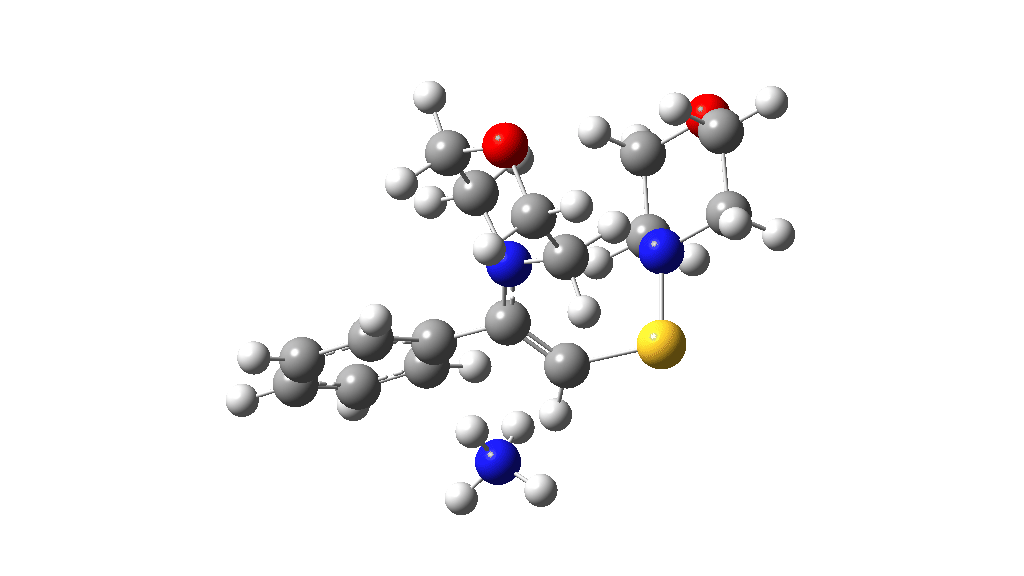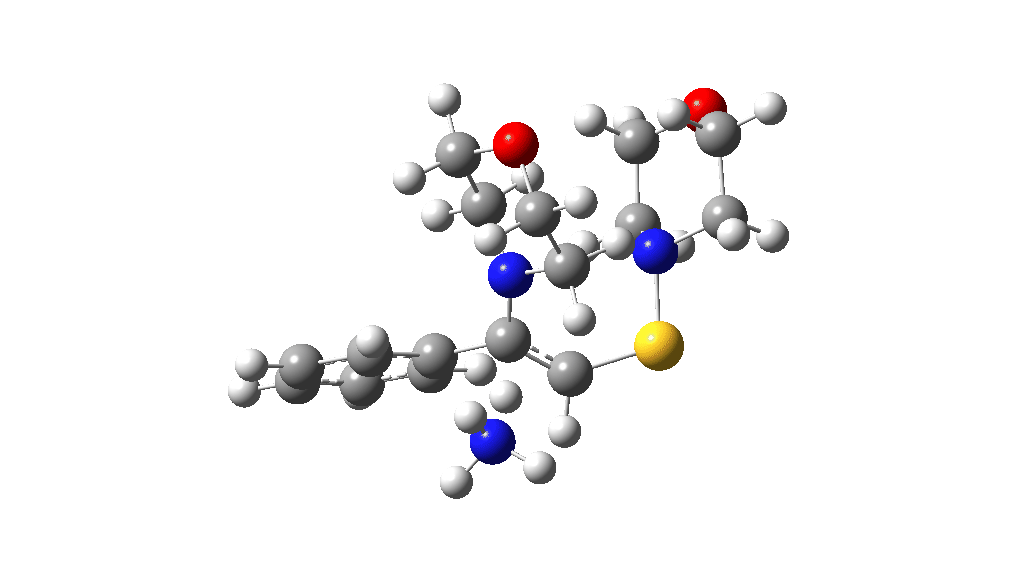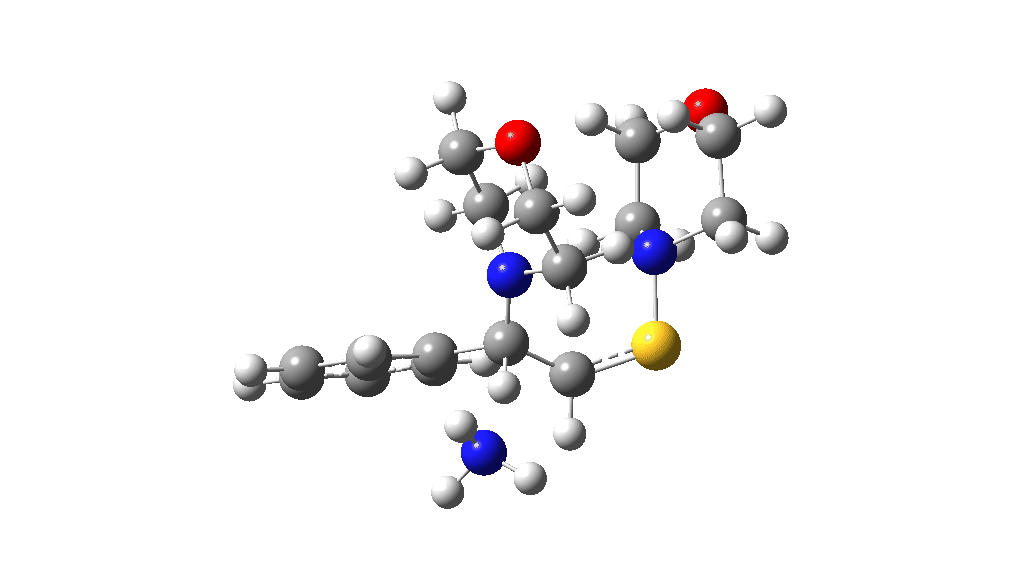
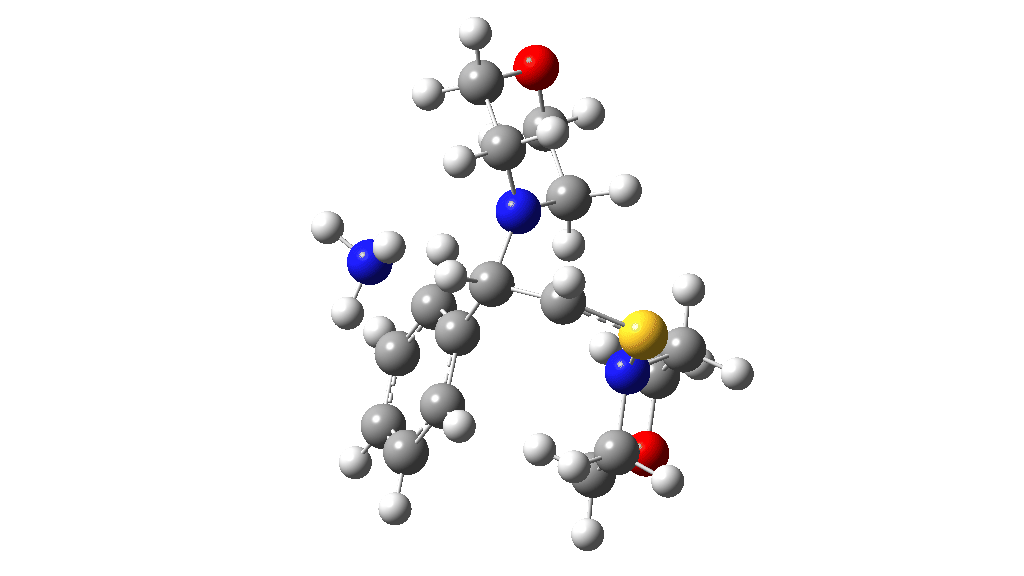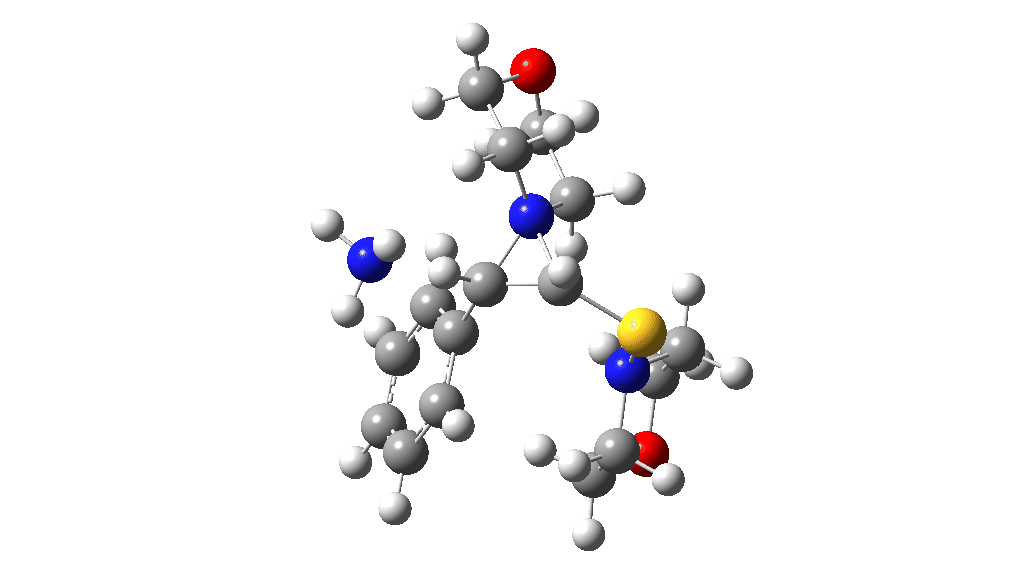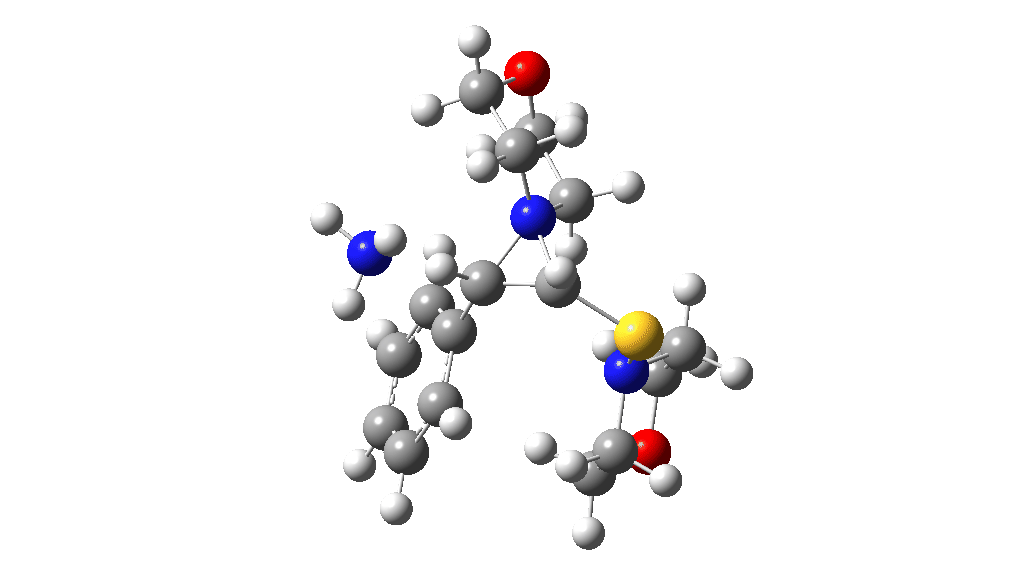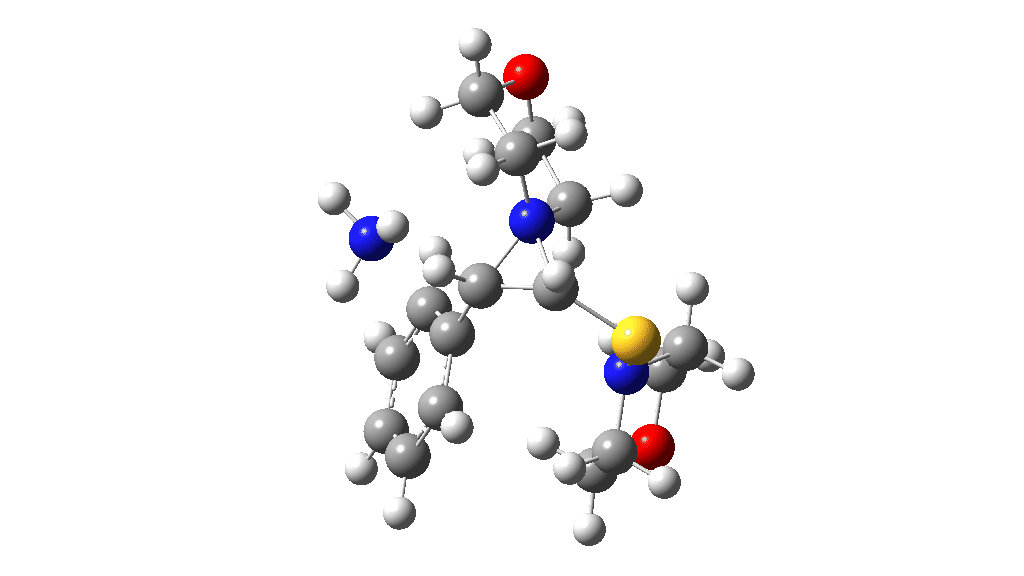
I noted above the process of optimising a transition state model by various refinements. Thus as a proton removal base, I originally used ammonia. Replacing this with morpholine as base has this effect. TS4: 38.2 → 36.0 kcal/mol and TS10: 36.9 → 33.6 kcal/mol. These are certainly in the right direction towards the expected barrier of ~30 ± 2 kcal/mol.