In this earlier post, I described how the stereochemistry of π2+π2 cycloadditions occurs suprafacially if induced by light, and how one antarafacial component appears if the reaction is induced by heat alone. I also noted how Woodward and Hoffmann (WH) explained that violations to their rules were avoided by mandating a change in mechanism requiring stepwise pathways with intermediates along the route. Here I illustrate how the stereochemistry of a thermal π2+π2 cycloaddition can indeed avoid an antarafacial component by performing appropriate gymnastic contortions instead of a mechanistic change (a WH violation certainly in the letter of their law, if not their spirit).
The IRC for the reaction (R=CH=S) shows the two molecules moving towards each other in a trapezoidal motion, with the axes of the C=C bonds parallel and the faces clearly suprafacial. At IRC ~-4, the nature of motion changes into a slide of one C=C axis along the other, converting the trapezoidal geometry into a square. By doing so, a full frontal “violation” is changed into a more subtle sideways insertion.
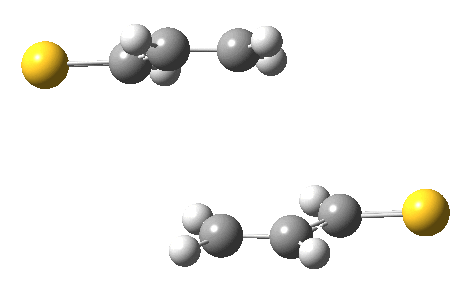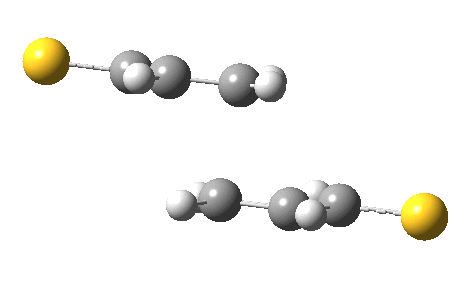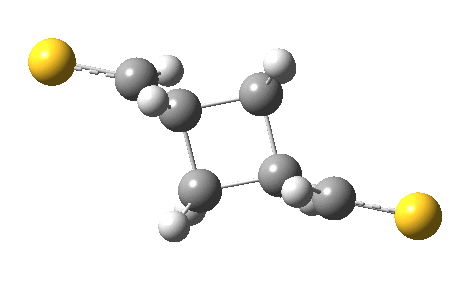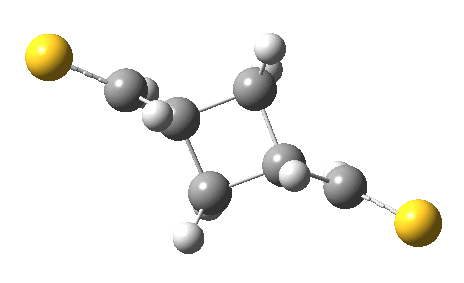
Animation of a 2+2 thermal cycloaddition with no antarafacial components. Click for 3D.
Tags: animation, pericyclic, Tutorial material
[…] Henry Rzepa Chemistry with a twist « Molecular gymnastics in 2+2 cycloadditions. […]
[…] But there is a (rarely cited or observed) alternative, as was illustrated for the π2+π2 cycloaddition of ethene to itself. There we saw the gymnastics of a limbo dancer, with one ethene sliding up to the other rather than […]
[…] asymmetric example is the 2+2 closed shell cycloaddition of two ethenes, which adopt a different form of […]
[…] bond, and one can normally rely on that bond length being in the range 2.0 – 2.3Å. Thus the thermal 2+2 cycloaddition of two ethenes can have a C-C length of 2.0Å, albeit accompanied by a fascinating trapezoidal geometry. My […]