Thus famously wrote Woodward and Hoffmann (WH) in their classic monograph about the conservation of orbital symmetry in pericyclic reactions. But they also note that the “fantastic” hydrocarbon (number 85 in their review) shown below presents a situation of great interest in having a half life of ~30 minutes at 353K (a free energy barrier of ~ 26.2 kcal/mol). Here I investigate if it might actually be such a violation.
I should first note that WH expect that violating reactions are likely to comport themselves via a non-concerted reaction path involving discrete intermediates.1 Which in the above case would be a biradical. But why is it an interesting example? Because, as a 4n (n=2) electron electrocyclic reaction (involving the bonds shown in red above), it must involve one antarafacial component. This is apparently rendered impossible (so WH claim) by the very rigid geometry of the system. However, an alternative, and geometrically more viable reaction involving only suprafacial components would indeed be be a violation according to their definition, if it were to be concerted, without (biradical) intermediates. So if a concerted pathway with no antarafacial components could be found, it would constitute a violation.
To model this system, the benzo groups (blue) are first removed. A transition state for the reaction is found [ωB97XD/6-311g(d)] with ΔG† 21.5 kcal/mol and an intrinsic reaction coordinate (IRC) that shows a concerted profile, albeit one with quite unusual features revealed in the gradient norm along the IRC.
- One might first note that the calculated barrier is similar to that measured for the real reaction (albeit with benzo groups). Although this does not prove that a lower energy process (such as involving biradicals) does not occur, it does at least suggest that the concerted pathway is not unreasonable given the observed kinetics of the reaction.
- The geometry up to the transition state (IRC=0.0 above) retains a plane of symmetry, and there is no hint of any axis of symmetry developing that might be associated with twisting due to an antarafacial component (click on the graphic below to inspect this geometry). However, the length of the cleaving bond (2.85Å) is unusually long for a transition state involving C-C cleavage, and the double bond (green above) is still intact (1.33Å). There is however an asymmetry developing, in which one of the 6-rings is moving faster than the other.
- At an IRC value of +5 (well past the transition state), something unexpected starts to happen; it is best seen as a very prominent feature in the gradient norm. Only now does the C=C bond start to lengthen to that typical of a pericyclic transition state (~1.40Å).
- By IRC +8, the erstwhile C=C bond has reached 1.43Å, but the geometry still retains most of its plane of symmetry. At IRC +10, this suddenly and abruptly breaks, and one trans alkene starts to form in the rhs ring. You can see this in the animation below, where one hydrogen suddenly accelerates its motion to fully assimilate the trans position (this phenomenon is a feature of so-called valley-ridge inflection points) and the other reverses its own motion. Prior to this, the trans component had been divided amongst two C=C bonds in the forming product, thus preserving the plane of symmetry.
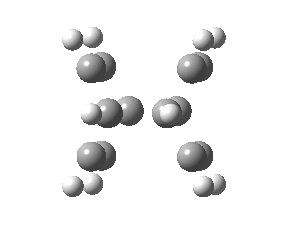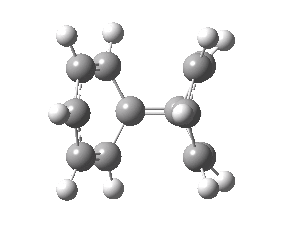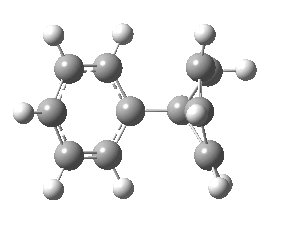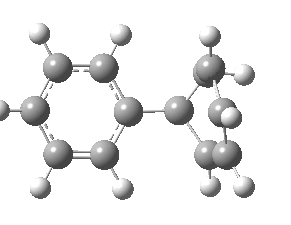
Animation of the geometry along the IRC. Click for 3D.
- The product of this IRC is thus a biphenyl where one of the phenyl rings sustains a trans component, a Möbius benzene in fact! Appropriately, the bond lengths in this (antiaromatic) ring alternate, whereas they are all equal in the other (aromatic) ring.
Click to invoke Jmol Rotate with mouse 
A Möbius benzene. Click for 3D
- Well, what a journey, in which most of the interesting action occurred AFTER the transitions state, controlled by the subsequent forces (dynamics) acting on the potential. It turned out to be a concerted reaction with a reasonable barrier. At the transition state itself, it was looking as if it might actually be a violation in the WH sense. But the requisite – better late than never – antarafacial component was indeed incorporated into the final product
WH argued that violations of their rules would be avoided by the reacting system adopting a stepwise, non-concerted pathway. It may be that the dynamics of reactions would also allow avoidance to occur by adopting concerted, but asynchronous geometrical distortions such as those seen here.2
1 “When I use a word,” Humpty Dumpty said in rather a scornful tone, “it means just what I choose it to mean – neither more nor less.” I add this quote, since the WH approach is based on an orbital picture deriving from a single determinantal SCF solution of the Schroedinger equation. In so-called multi-configurational treatments (MCSCF), molecular orbitals for a single configuration no longer occupy such a central position in the theory.
2 For example, it could be argued that a violation of the WH rules for 2πs + 2πs thermal cycloadditions can be avoided by a trapezoidal distortion, DOI: 10.1039/A805668D.
Tags: electrocyclic reaction, Historical, pericyclic, Tutorial material
[…] by light, and how one antarafacial component appears if the reaction is induced by heat alone. I also noted how Woodward and Hoffmann (WH) explained that violations to their rules were avoided by mandating a […]
[…] left the story of the molecule below on the precipice of a cliff. I had shaved off the four benzo groups (blue) in […]