A pyrophoric metal is one that burns spontaneously in oxygen; I came across this phenomenon as a teenager doing experiments at home. Pyrophoric iron for example is prepared by heating anhydrous iron (II) oxalate in a sealed test tube (i.e. to 600° or higher). When the tube is broken open and the contents released, a shower of sparks forms. Not all metals do this; early group metals such as calcium undergo a different reaction releasing carbon monoxide and forming calcium carbonate and not the metal itself. Here as a prelude to the pyrophoric reaction proper, I take a look at this alternative mechanism using calculations.
There are ~60 crystal structures of metal oxalates, of which several are naturally occurring minerals (Fe, humboldtine[1], Ca, Weddellite[2], Li[3], Na[4], K[5], Cs[6]. The natural geometry of the oxalate di-anion is planar (torsion 0 or 180°) but a small number are twisted such as the caesium oxalate.
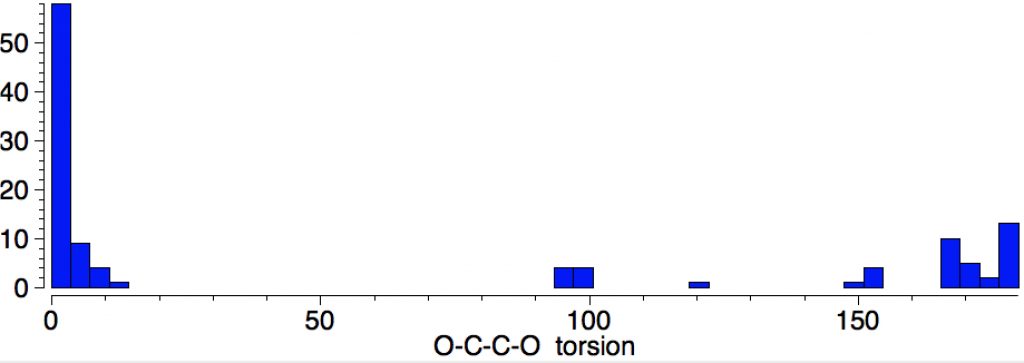
The kinetics of pyrolysis of a number of metal oxalates were studied some years ago (Ca[7], Li[8]) indicating barriers ranging from 53-68 kcal/mol. One proposed mechanism is as shown in this article.[7]
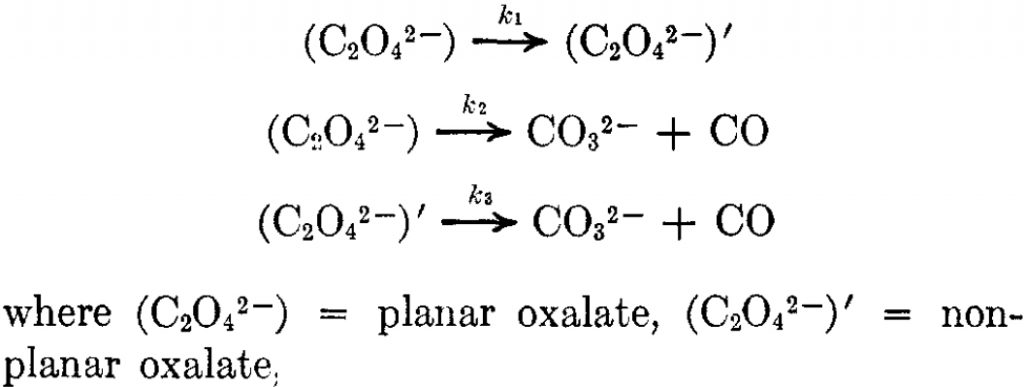
It was surmised from the kinetic analysis that the k1 activation step (rotation about the C-C bond from planar to twisted) was ~12 ± 20 kcal/mol, whilst steps k2 or k3 had the much higher activation energy noted above. A search (of Scifinder) for quantum mechanical “reality checks” of this mechanism revealed a blank and so I apply such a check here using Mg as the metal.
The carbonyl extrusion step (ωB97XD/Def2-TZVPPD/SCRF=water, DOI: 10.14469/hpc/2320) was studied with a water solvent field applied in an effort to mimic the solid state crystal structure of the species as a better representation of the ionic lattice than a pure vacuum calculation.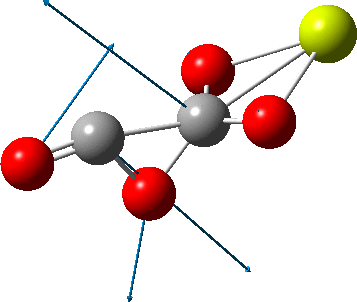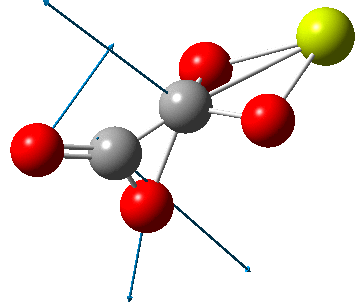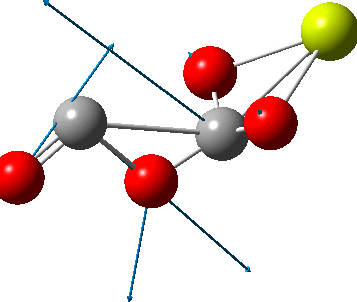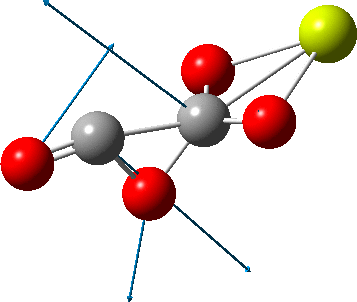
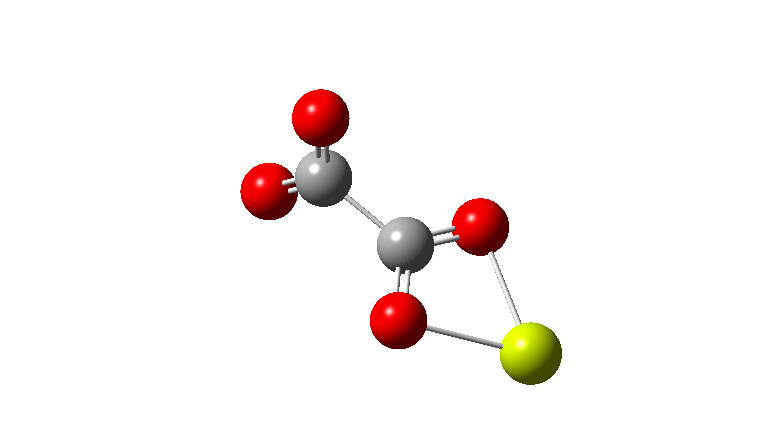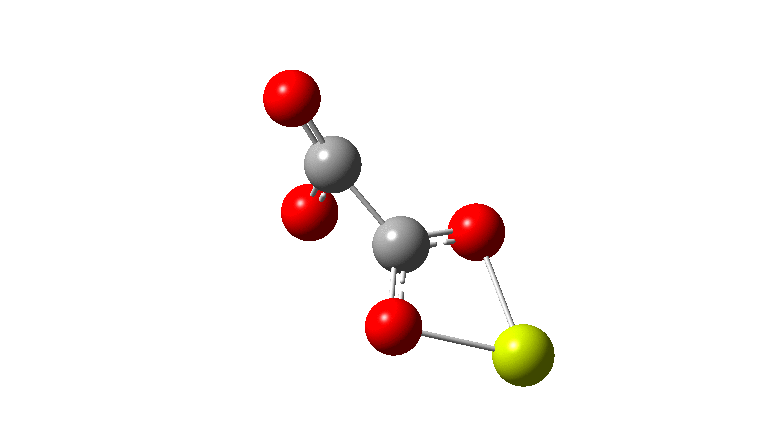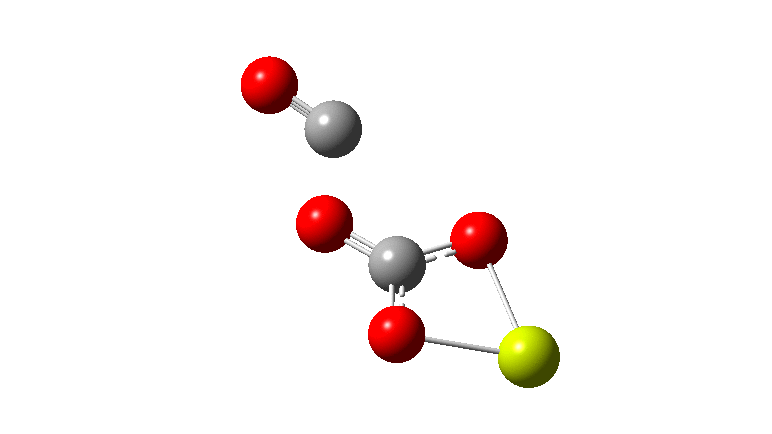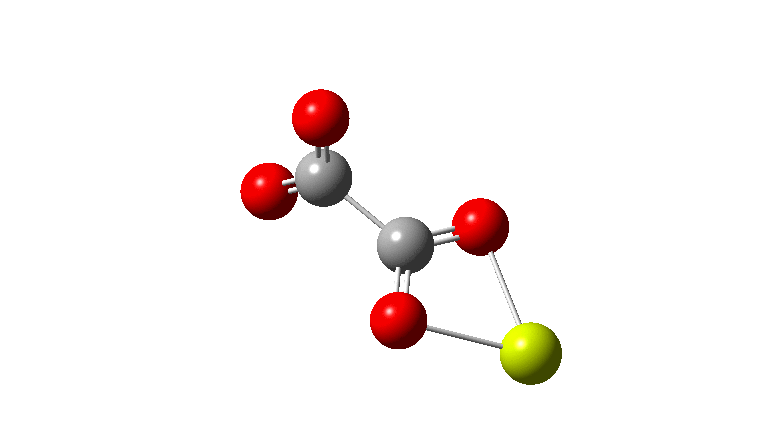 An IRC (intrinsic reaction coordinate, DOI: 10.14469/hpc/2324) reveals the start-point geometry still has a very small negative force constant (-38 cm-1, DOI: 10.14469/hpc/2321) which now corresponds to a small rotation about the C-C bond to give a C2-symmetric conformation:
An IRC (intrinsic reaction coordinate, DOI: 10.14469/hpc/2324) reveals the start-point geometry still has a very small negative force constant (-38 cm-1, DOI: 10.14469/hpc/2321) which now corresponds to a small rotation about the C-C bond to give a C2-symmetric conformation:
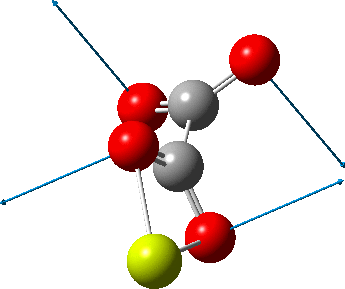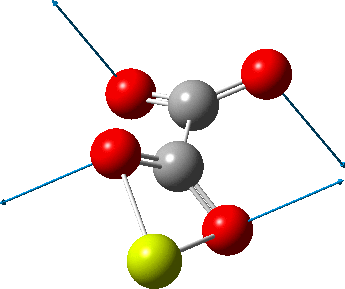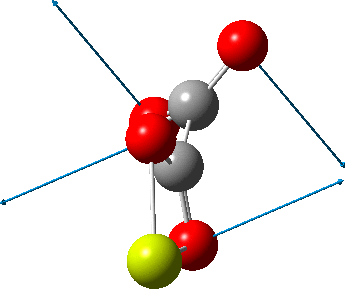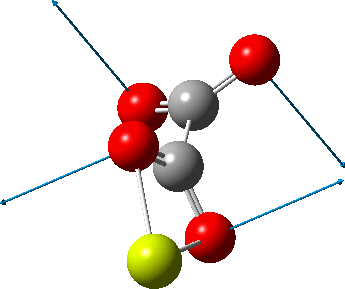 But the barrier for this process is tiny and nothing like the ~12 ± 20 kcal/mol inferred from the kinetic analysis. Perhaps most of the incentive to pack into a totally planar geometry comes from the interactions in the ionic lattice. The calculated free energy barrier (ΔG298‡ 54.7 kcal/mol, ΔG755‡ 55.1 kcal/mol) is within the reported measured range.
But the barrier for this process is tiny and nothing like the ~12 ± 20 kcal/mol inferred from the kinetic analysis. Perhaps most of the incentive to pack into a totally planar geometry comes from the interactions in the ionic lattice. The calculated free energy barrier (ΔG298‡ 54.7 kcal/mol, ΔG755‡ 55.1 kcal/mol) is within the reported measured range.
The mechanism for production of pyrophoric metal itself is likely to be far more complex, involving (inter alia) electron transfer from oxygen to metal. If I find anything I will report back here.
References
- T. Echigo, and M. Kimata, "Single-crystal X-ray diffraction and spectroscopic studies on humboldtine and lindbergite: weak Jahn–Teller effect of Fe2+ ion", Physics and Chemistry of Minerals, vol. 35, pp. 467-475, 2008. https://doi.org/10.1007/s00269-008-0241-7
- C. Sterling, "Crystal structure analysis of weddellite, CaC2O4.(2+x)H2O", Acta Crystallographica, vol. 18, pp. 917-921, 1965. https://doi.org/10.1107/s0365110x65002219
- G.A. Jeffrey, and G.S. Parry, "The Crystal Structure of Sodium Oxalate", Journal of the American Chemical Society, vol. 76, pp. 5283-5286, 1954. https://doi.org/10.1021/ja01650a007
- Dinnebier, R.E.., Vensky, S.., Panthofer, M.., and Jansen, M.., "CCDC 192180: Experimental Crystal Structure Determination", 2003. https://doi.org/10.5517/cc6fzcy
- Dinnebier, R.E.., Vensky, S.., Panthofer, M.., and Jansen, M.., "CCDC 192182: Experimental Crystal Structure Determination", 2003. https://doi.org/10.5517/cc6fzf0
- F.E. Freeberg, K.O. Hartman, I.C. Hisatsune, and J.M. Schempf, "Kinetics of calcium oxalate pyrolysis", The Journal of Physical Chemistry, vol. 71, pp. 397-402, 1967. https://doi.org/10.1021/j100861a029
- D. Dollimore, and D. Tinsley, "The thermal decomposition of oxalates. Part XII. The thermal decomposition of lithium oxalate", Journal of the Chemical Society A: Inorganic, Physical, Theoretical, pp. 3043, 1971. https://doi.org/10.1039/j19710003043
Tags: Aluminium, calculated free energy barrier, Carbon monoxide, Chemical elements, Chemistry, higher activation energy, Iron, Matter, metal, metal oxalates, Oxide, pyrophoric metal, Pyrophoricity, Reducing agents
Here is a very preliminary look at the alternative mechanism, resulting in a metal atom and two molecules of carbon dioxide. An initial model might be the triplet state of the metal salt, in which one electron is promoted from the oxalic acid component to the empty 3s shell of the metal, resulting in an ion-pair in which the C-C length is substantially longer than a single bond.
This triplet is computed (DOI: 10.14469/hpc/2327) to be 21.5 kcal/mol higher in free energy than the transition state for carbonate formation via oxygen transfer. This corresponds to the lack of any substantial observed CO2 pathway for this type of early period metal.