The reaction between a carbene and an alkene to form a cyclopropane is about as simple a reaction as one can get. But I discussed before how simple little molecules (cyclopropenyl anion) can hold surprises. So consider this reaction:
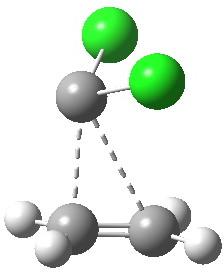
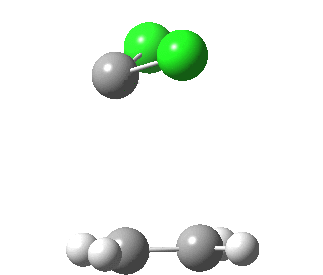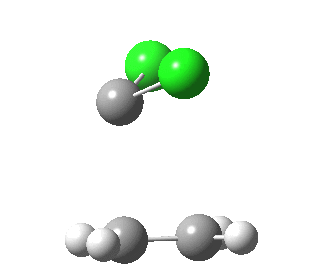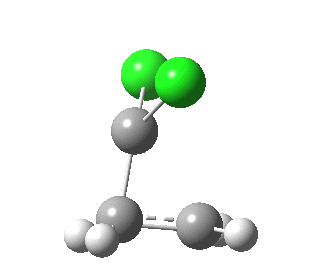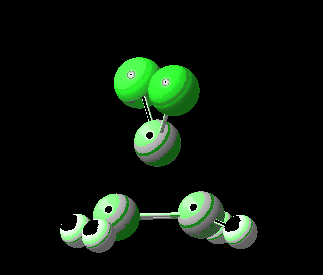
Transition state for reaction between ethene and dichlorocarbene. Click for 4D.
The reaction is a 4-electron pericyclic process, and so is subject to the Woodward-Hoffmann rules, which imply that such a 4n-thermal process should go with one antarafacial component. But there is a (rarely cited or observed) alternative, as was illustrated for the π2+π2 cycloaddition of ethene to itself. There we saw the gymnastics of a limbo dancer, with one ethene sliding up to the other rather than taking a full-frontal approach. But whilst that reaction had an unrealistic activation barrier of ~50 kcal/mol, the reaction between dichlorocarbene and an alkene is known to be a very facile one. And so the calculation shows (below). The barrier to reaction is small, and so this is an example of a low-barrier nominally forbidden reaction which nevertheless achieves a low barrier by avoiding the direct approach of the two molecules and adopting a round-about path!
 |
|
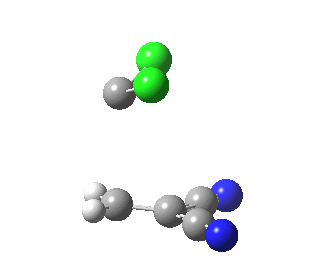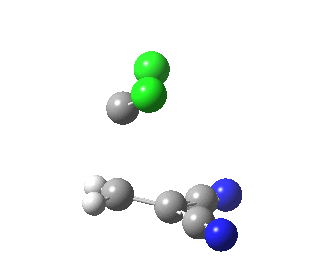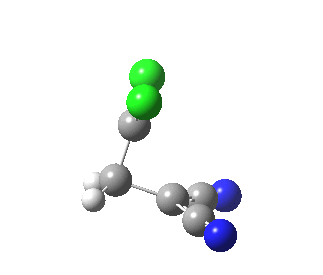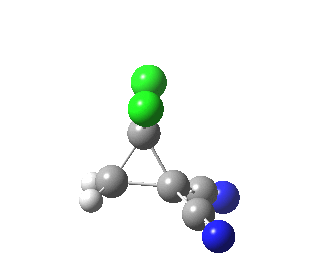 |
This round-about approach is seen best in the IRC for the addition to dicyano-ethene. Shown above is the gradient norm along the IRC.
- From IRC -1.2 to 0.0 (the transition state) the reaction corresponds to the formation of effectively just one C-C bond (a two electron process if you like).
- At IRC +2.0 a second distinct feature is seen in the graph, and this now corresponds to the formation of the second C-C bond, involving a sliding motion of the carbene (again, a two-electron process).
So by breaking a four-electron process into two phases, each involving just one electron pair, a lot of the forbidden Woodward-Hoffmann character seems to be avoided. Truly the direct approach not being the best!
Tags: limbo dancer, pericyclic, Reaction Mechanism, Tutorial material
[…] was that the initial attack of oxygen on the alkyne was very asymmetric. This reminded of another post on the reaction of dichlorocarbene with ethene, which too is asymmetric, yet again to avoid an […]
[…] elimination) of dichlorocarbene to ethene is a classic example of a forbidden pericyclic process taking a roundabout route to avoid directly violating the Woodward-Hoffmann rules. However, a thermal six-electron process […]
[…] addition of dichlorocarbene to ethene (see this blog for […]