Each year, C&E News runs a poll for their “Molecule of the year“. I occasionally comment with some aspect of one of the molecules that catches my eye (I have already written about cyclo[18]carbon, another in the list). Here, it is the Incredible chloride cage, a cryptand-like container with an attomolar (1017 M-1) affinity for a chloride anion.[1] The essence of the binding is six short CH…Cl– and one slightly longer interactions to the same chloride (DOI: 10.5517/ccdc.csd.cc1ngqrl) and one further hydrogen bond to a water molecule; eight coordinated chloride anion!
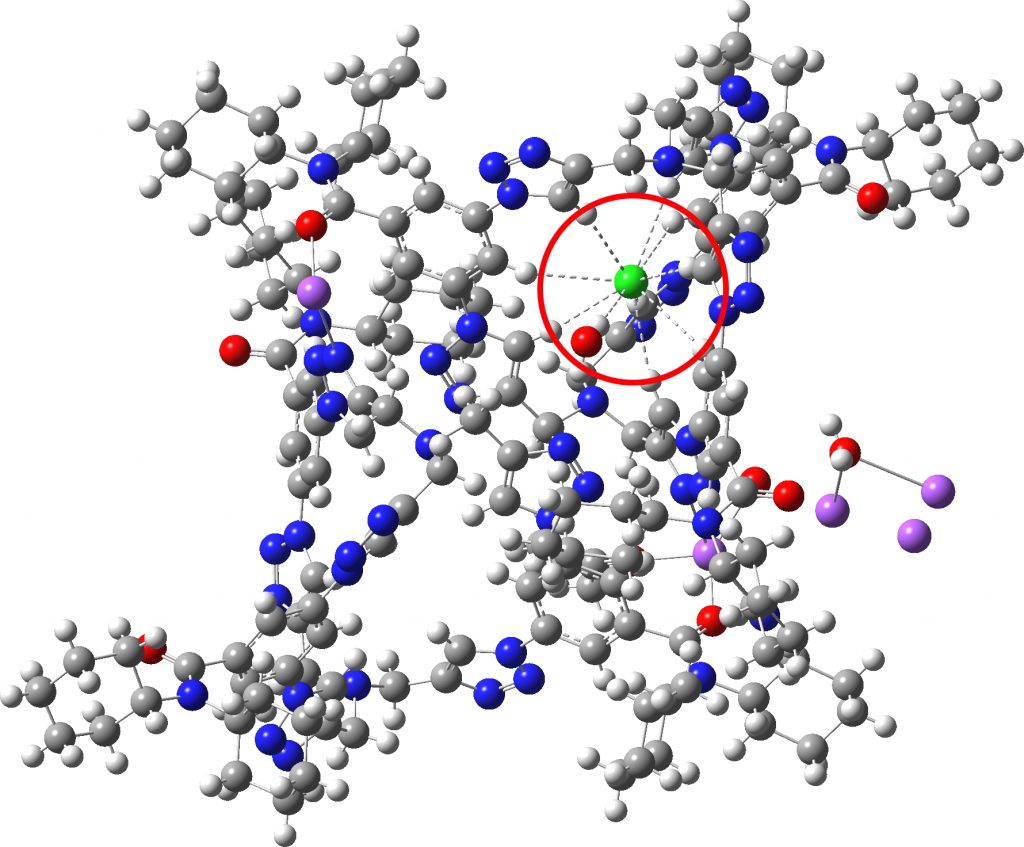
Click image for 3D model
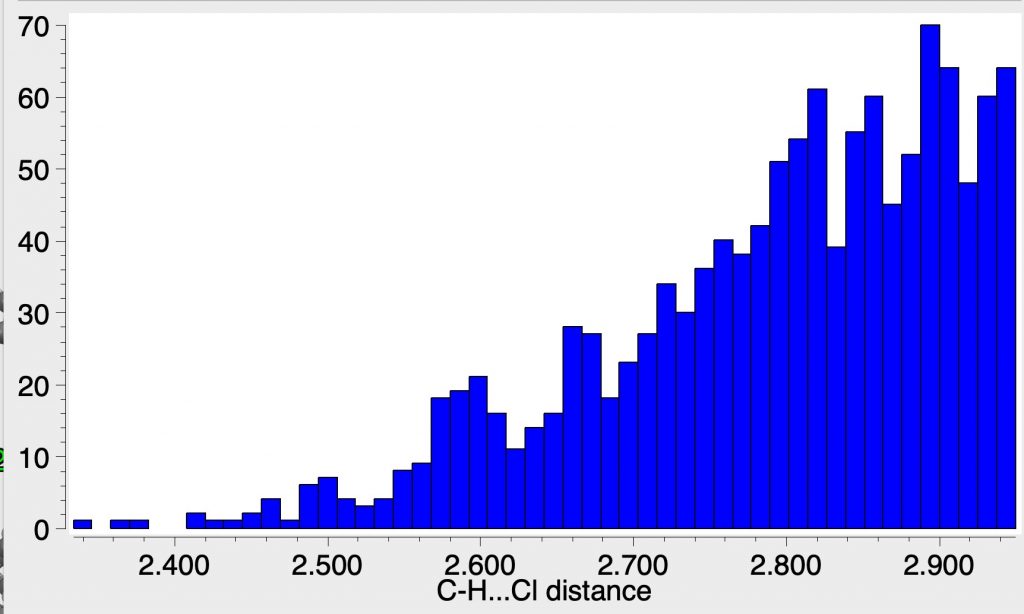
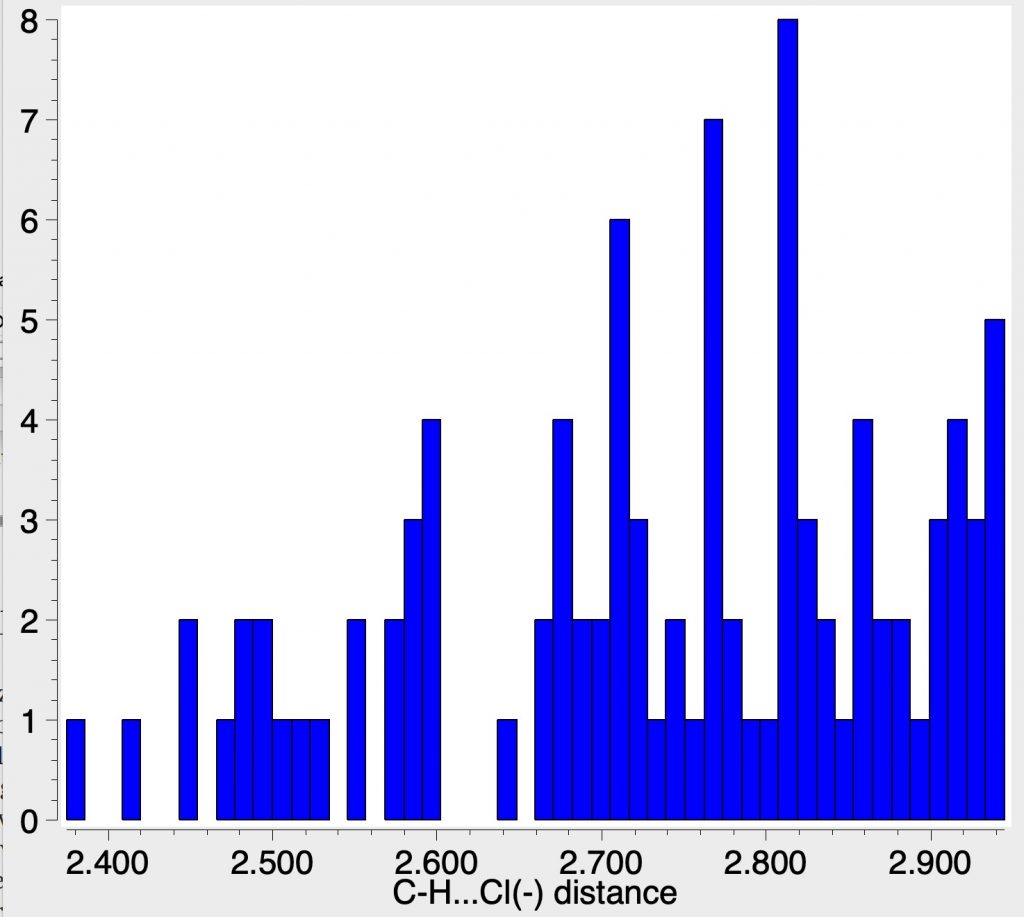
Here I thought I might explore how common the C-H…C– motif is in crystal structures by showing some searches of the CSD. The CH—Cl distances reported in the article are around 2.7-2.9Å and I wanted to see how these compare with other structures. The searches were done with the constraint of data collection <90K, to minimise vibrational noise and intermolecular contacts only.
The first search is for all contacts to Cl, including covalent chlorine and also perchlorate anions, and you can see that they can extend down to about 2.4Å, although the commonest short contact is 2.5Å
This second search is for chloride anion only (by excluding any higher coordination at that atom), and the distribution does not really change.
This final search is for any chloride anion with at least THREE CH…Cl contacts to the same atom and now you are seeing ~2.7Å as the commonest short contact, which corresponds exactly with the molecule reported above.
So the apparently weak CH…Cl hydrogen bond, if there are seven of them, can accumulate to give attomolar binding affinities. Wow, imagine a drug with that sort of affinity!
References
- Y. Liu, W. Zhao, C. Chen, and A.H. Flood, "Chloride capture using a C–H hydrogen-bonding cage", Science, vol. 365, pp. 159-161, 2019. https://doi.org/10.1126/science.aaw5145
Roald Hoffmann has pointed me to DOI: 10.1073/pnas.1615742114 which contains an eight-coordinate fluoride anion in a silicate cage. It is also cited as an interesting example of hypervalence (or is it hypercoordination?)