In my first post on the topic, I discussed how inverting the polarity of the C-X bond from X=O to X=Be (scheme below) could flip the stereochemical course of the electrocyclic pericyclic reaction of a divinyl system. This was followed up by exploring what happens at the half way stage, i.e. X=CH2, the answer being that one gets an antarafacial pathway as with X=O. Here I fill in another gap, X=BH to see if a metaphorical microscope can be used to view the actual region of the “flip” to a suprafacial mode. This time, uniquely, it proved possible to locate TWO transition states for this process, one suprafacial[1] and one antarafacial[2], this latter being 10.5 kcal/mol lower in ΔG† (ωB97XD/6-311G(d,p)/SCRF=dichloromethane). It is quite rare to be able to find BOTH stereochemical outcomes of a thermal pericyclic reaction.‡
First, the antarafacial IRC (X=BH)[3]. There are several interesting features. Note at IRC = -8, the divinyl compound appears as a Hidden Intermediate (HI), having formed from a compound where the HB=C substituent has ring opened from a cyclobutene-like precursor (initial electrocyclic). If you watch the animation, you can see the antarafacial bond forming from the bottom face of the vinyl group on the left to the top face of the vinyl group in the HI on the right (antarafacial=conrotation). Because the entire process is concerted (no real intermediates participate), we have here an unusual pericyclic cascade where one electrocyclic reaction is immediately followed by another quite different one.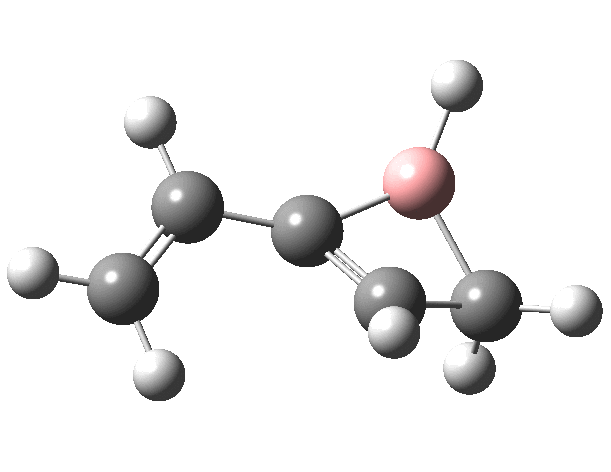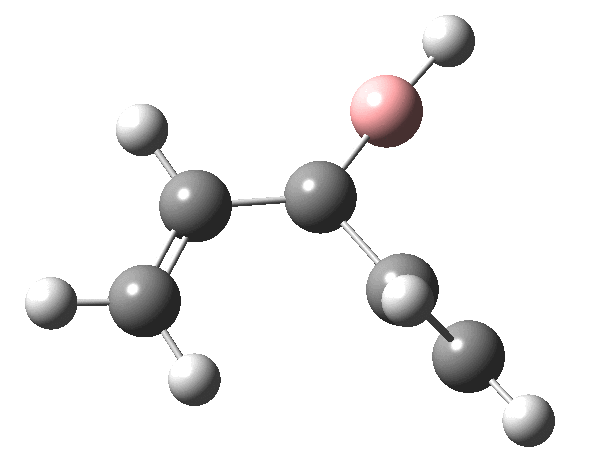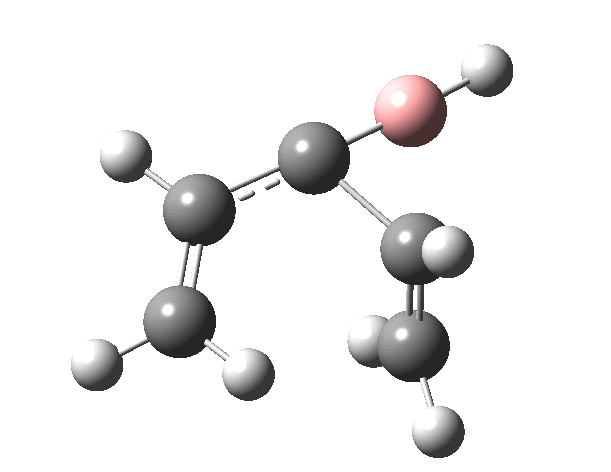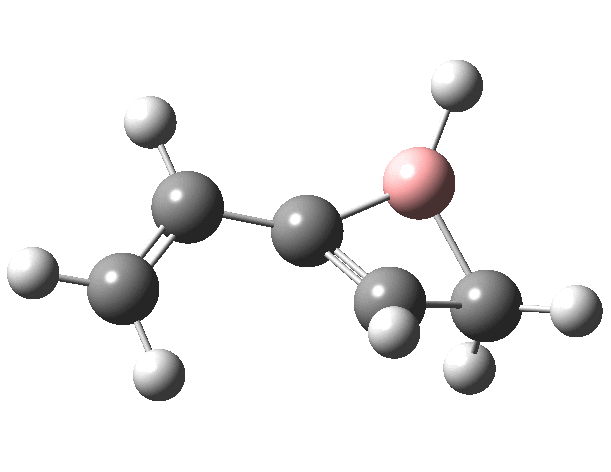
Now for the suprafacial IRC[4]. It is pretty similar to the previous path, but again if you inspect very carefully you will see that it is the TOP face of the vinyl group on the left forming the bond to the TOP face of the vinyl group on the right (suprafacial/disrotation).
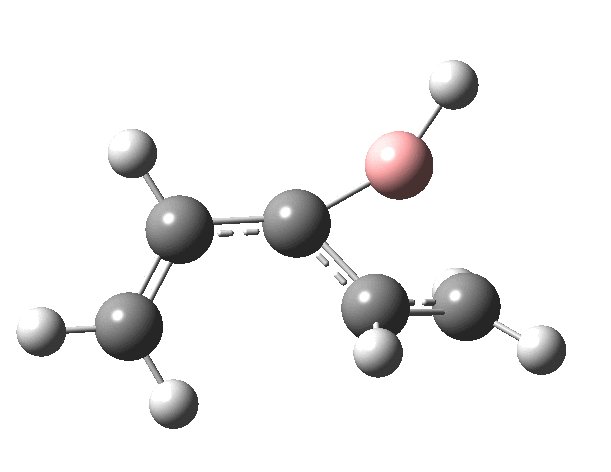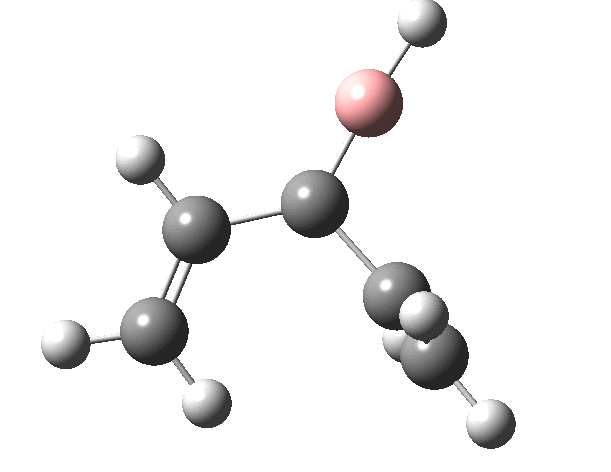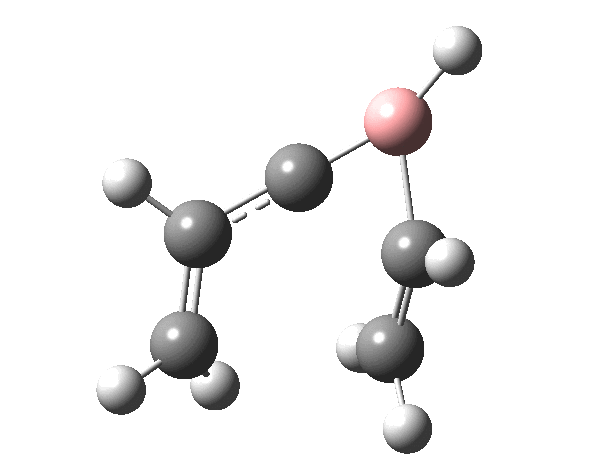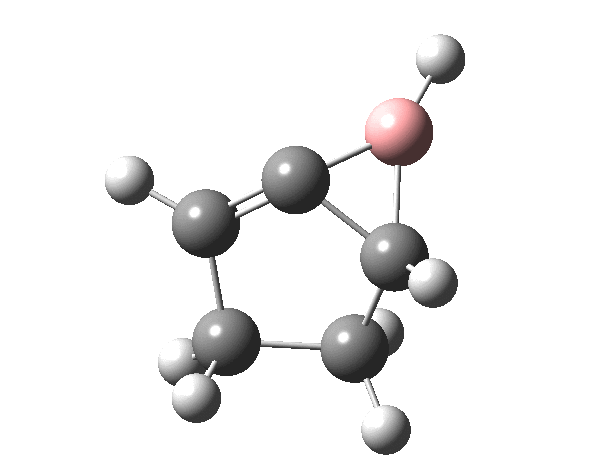
You might ask if the molecules used here are realistic, i.e. could they form the basis of real reactions to be conducted in a laboratory? Well, the C=B-C fragment has 9 hits in the CCDC crystal database (none for C=B-H). One example is cited here.[5]. So, yes, possibly a realistic system, except the barriers do look too high. Perhaps suitable substituents might help? But even if this could not be carried out in a test-tube, it does teach one about pericyclic reactions and how one might manipulate them.
‡One such is the [1,6] sigmatropic shift in homotropylium cation involving migration of a Me2C+ group, where the “allowed” process in which the migrating group retains its configuration has a barrier of 17.7 kcal/mol and the “forbidden” route where the migrating group inverts its configuration with a barrier of 38.6 kcal/mol (thanks to Alex Genaev; I will strive to make the coordinates available via the repository shortly).
References
- H.S. Rzepa, "Gaussian Job Archive for C5H7B", 2014. https://doi.org/10.6084/m9.figshare.1133933
- H.S. Rzepa, "Gaussian Job Archive for C5H7B", 2014. https://doi.org/10.6084/m9.figshare.1133934
- H.S. Rzepa, "Gaussian Job Archive for C5H7B", 2014. https://doi.org/10.6084/m9.figshare.1133936
- H.S. Rzepa, "Gaussian Job Archive for C5H7B", 2014. https://doi.org/10.6084/m9.figshare.1134015
- M. Menzel, H.J. Winkler, T. Ablelom, D. Steiner, S. Fau, G. Frenking, W. Massa, and A. Berndt, "Diborylcarbenes as Reactive Intermediates in Double 1,2‐Rearrangements with Low Activation Enthalpies", Angewandte Chemie International Edition in English, vol. 34, pp. 1340-1343, 1995. https://doi.org/10.1002/anie.199513401
Tags: Alex Genaev, Hawaii, HB