The previous post explored why E2 elimination reactions occur with an antiperiplanar geometry for the transition state. Here I have tweaked the initial reactant to make the overall reaction exothermic rather than endothermic as it was before. The change is startling.
The exothermicity is of course due to the aromatisation of the ring. The IRC is however quite different from before.
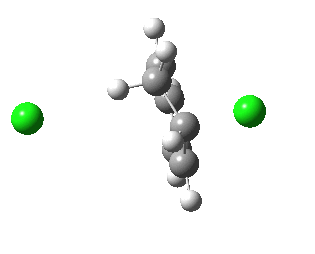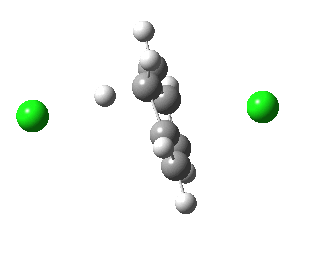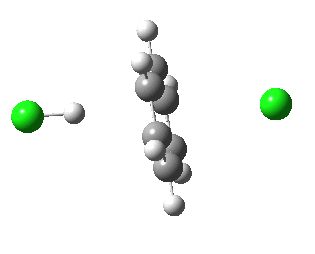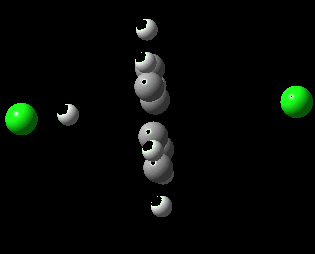
IRC for E2 elimination. Click for 3D
- The transition state (IRC=0.0) is reached early, and the initial movement is of the chlorine on the right. This in fact resembles an E1 elimination to form an intermediate carbocation. This, being a resonance stabilised (Wheland) type of cation, is particularly favoured.
- At this point, the H-C bond has scarcely started breaking (1.13Å).
- When IRC of -5 is reached, the C-Cl bond is essentially broken (3.05Å). At this point the energy is still higher than the reactant.
- A sudden abrupt change occurs resulting from rapid proton transfer at IRC -6, and the energy plummets to become exothermic.
So, like the SN1 reaction discussed in another post, this E2 reaction occurs in distinct stages, the first resembling an E1 mechanism, followed by a second phase leading to elimination. It is still a concerted reaction, but the proton transfer occurs only AFTER the transition state. The simple designation E2/E1 is clearly not a fully adequate description of such mechanisms.