The story so far. Inspired by the report of the most polar neutral compound yet made, I suggested some candidates based on the azulene ring system that if made might be even more polar. This then led to considering a smaller π-analogue of azulene, m-benzyne. Here I ponder how a derivative of this molecule might be made, using computational profiling as one reality check.
One reaction as envisaged is to prepare the penta-substituted benzene as shown below.
Abstraction of the proton using strong base might result in 1,3-elimination of the leaving group X to form the m-benzyne. For the substituent X=Cl, a ωB97XD/Def2-TZVPP/SCRF=thf calculation (DOI: 10.14469/hpc/2057) of the reaction profile shows a relatively large barrier to elimination, with an endothermic product.
A better leaving group might be X=OTf. Unlike, X=Cl, this reaction is nicely exoenergic (ΔΔG -10.6 kcal/mol) with an accessible activation free energy of 22.5 kcal/mol (DOI: 10.14469/hpc/2066, 10.14469/hpc/2123, 10.14469/hpc/2096)
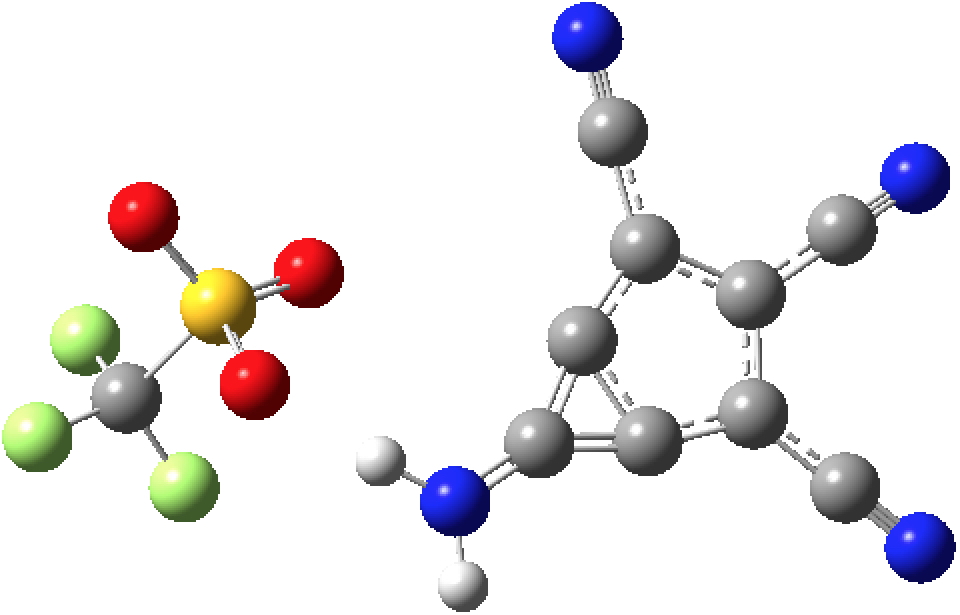
Perhaps then this, the smaller homologue of azulene, might indeed be capable of synthesis?
Tags: accessible activation free energy, Aryne, Azulenes, endothermic product
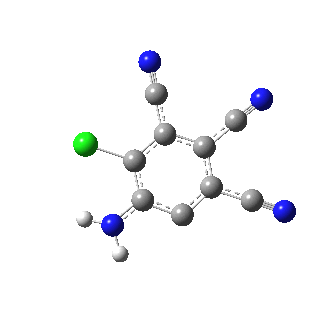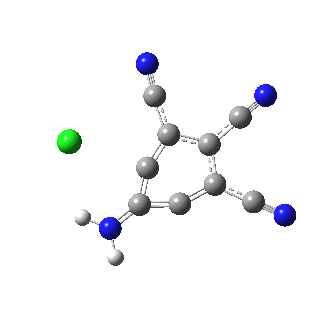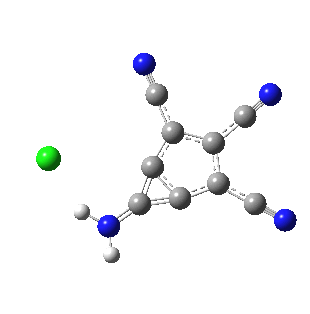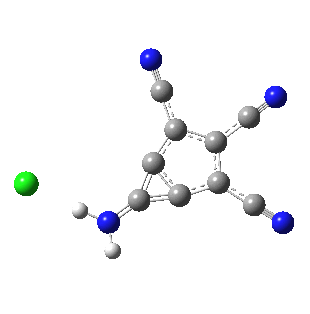
I like this idea! It would be wonderful to see such a molecule. Azulene is on my mind now as my organic class just completed their literature search for their spring “research project” – trying to make 1-methylazulene. I thought it would be fun for them to make a blue liquid while learning about aromaticity!
Would it be possible to make the chlorine analogue of your molecule, from tetra or pentachloroaniline or its N,N-dimethyl equivalent? These are commercially available or easily made. I can imagine that ortho lithiation of one of those might lead to your proposed structure, provided that chlorine can provide enough stabilisation.
Thanks for your many interesting posts!
Martin
I apologize for missing (in my excitement!) your mention that the reaction requires something like OTf as a leaving group, and Cl will probably fail. I did find one paper that you may find relevant, though: D. J. Berry, I. Collins, S. M. Roberts, H. Suschitzky and B. J. Wakefield J. Chem. Soc. C, 1969, 1285-1294. (DOI: 10.1039/J39690001285) The paper mentions lithiation as I proposed gives mixtures; it also describes some more conventional benzyne chemistry.
I’ll certainly ponder this to see if there is a viable route. Thanks again!
Martin A. Walker