In 2012, I wrote a story of the first ever reaction curly arrows, attributed to Robert Robinson in 1924. At the time there was a great rivalry between him and another UK chemist, Christopher Ingold, with the latter also asserting his claim for their use. As part of the move to White City a lot of bookshelves were cleared out from the old buildings in South Kensington, with the result that yesterday a colleague brought me a slim volume they had found entitled The Journal of the Imperial College Chemical Society (Volume 6).‡
Posts Tagged ‘Henry Armstrong’
Early “curly” (reaction) arrows. Those of Ingold in 1926.
Wednesday, August 22nd, 2018Benzene: an oscillation or a vibration?
Wednesday, May 28th, 2014In the preceding post, a nice discussion broke out about Kekulé’s 1872 model for benzene.[1] This model has become known as the oscillation hypothesis between two extreme forms of benzene (below). The discussion centered around the semantics of the term oscillation compared to vibration (a synonym or not?) and the timescale implied by each word. The original article is in german, but more significantly, obtainable only with difficulty. Thus I cannot access[1] the article directly since my university does not have the appropriate “back-number” subscription.‡ So it was with delight that I tracked down an English translation in a journal that I could easily access.[2] Here I discuss what I found (on pages 614-615, the translation does not have its own DOI).
References
- A. Kekulé, "Ueber einige Condensationsproducte des Aldehyds", Justus Liebigs Annalen der Chemie, vol. 162, pp. 77-124, 1872. https://doi.org/10.1002/jlac.18721620110
- "Organic chemistry", Journal of the Chemical Society, vol. 25, pp. 605, 1872. https://doi.org/10.1039/js8722500605
Henry Armstrong: almost an electronic theory of chemistry!
Monday, November 7th, 2011Henry Armstrong studied at the Royal College of Chemistry from 1865-7 and spent his subsequent career as an organic chemist at the Central College of the Imperial college of Science and technology until he retired in 1912. He spent the rest of his long life railing against the state of modern chemistry, saving much of his vitriol against (inter alia) the absurdity of ions, electronic theory in chemistry, quantum mechanics and nuclear bombardment in physics. He snarled at Robinson’s and Ingold’s new invention (ca 1926-1930) of electronic arrow pushing with the put down “bent arrows never hit their marks“.‡ He was dismissed as an “old fogy, stuck in a time warp about 1894.”‡ So why on earth would I want to write about him? Read on…
The oldest reaction mechanism: updated!
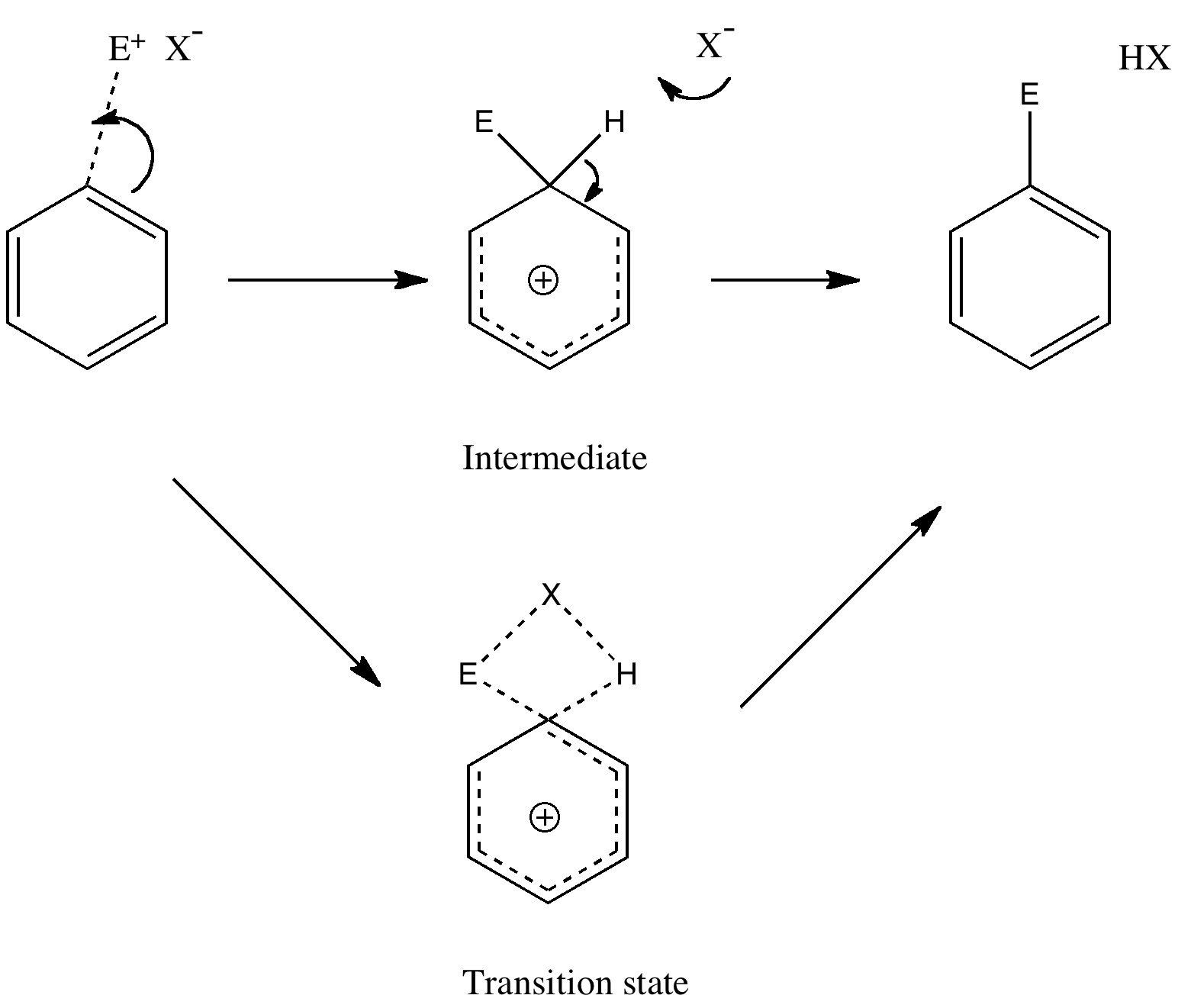
Tuesday, September 14th, 2010Unravelling reaction mechanisms is thought to be a 20th century phenomenon, coincident more or less with the development of electronic theories of chemistry. Hence electronic arrow pushing as a term. But here I argue that the true origin of this immensely powerful technique in chemistry goes back to the 19th century. In 1890, Henry Armstrong proposed what amounts to close to the modern mechanism for the process we now know as aromatic electrophilic substitution [1]. Beyond doubt, he invented what is now known as the Wheland Intermediate (about 50 years before Wheland wrote about it, and hence I argue here it should really be called the Armstrong/Wheland intermediate). This is illustrated (in modern style) along the top row of the diagram.
References
- "Proceedings of the Chemical Society, Vol. 6, No. 85", Proceedings of the Chemical Society (London), vol. 6, pp. 95, 1890. https://doi.org/10.1039/pl8900600095
Clar islands in a π Cloud
Wednesday, December 9th, 2009Clar islands are found not so much in an ocean, but in a type of molecule known as polycyclic aromatic hydrocarbons (PAH). One member of this class, graphene, is attracting a lot of attention recently as a potential material for use in computer chips. Clar coined the term in 1972 to explain the properties of PAHs, and the background is covered in a recent article by Fowler and co-workers (DOI: 10.1039/b604769f). The concept is illustrated by the following hydrocarbon: