If you have not previously visited, take a look at Nick Greeves’ ChemTube3D , an ever-expanding gallery of reactions and their mechanisms. The 3D is because all molecules are offered with X, Y and z coordinates. You also get arrow pushing‡ in 3D. Here, I argue that we should adopt Einstein, and go to the space-time continuum! By this, I mean one must also include the order in which things happen. To my knowledge, no compendium of (organic) reaction mechanisms incorporates this 4th dimension. My prelude to this post nicely illustrated this latter aspect. Here I continue with an exploration of the mechanism of forming an acyl chloride from a carboxylic acid using thionyl chloride. The mechanism shown at ChemTube3D is as below and will now be tested for its reasonableness using quantum mechanics.
Step (a, R=Me) is shown below (ωB97XD/6-311G(d,p)/SCRF=acetic acid);
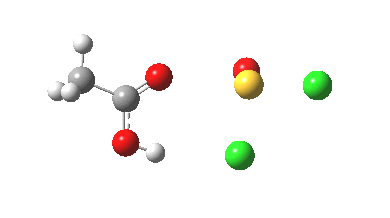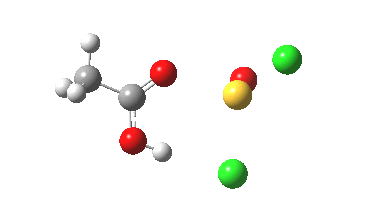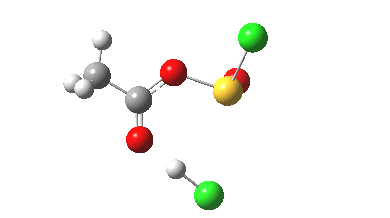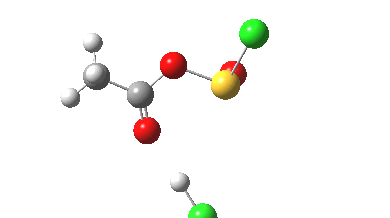
- From IRC -6 to -2, the oxygen of the acid carbonyl approaches the sulfur.
- IRC -2 then shows one chlorine to start move towards the OH, and the sulfur now adopts a “figure T” coordination.
- By IRC +2, the O…H…Cl angle has become almost linear, which is the optimum geometry for a proton transfer
- At IRC +3, the proton transfer from O to Cl is about half complete…
- A process largely complete by IRC +4.5
- Some residual activity takes place on the methyl group, which reorients itself with respect to the adjacent C-O bonds.
- The free energy barrier ΔG is 21.9 kcal/mol, which perhaps might be lowered if a solvation model including explicit hydrogen bonds were to be used.
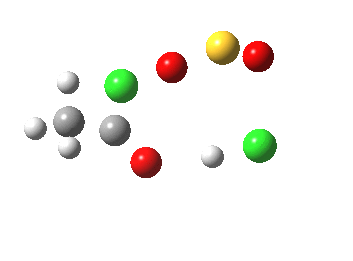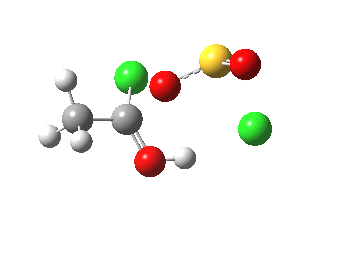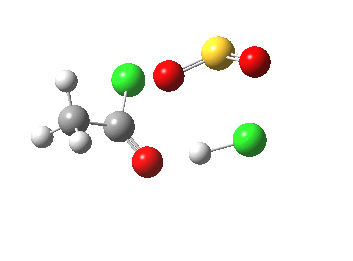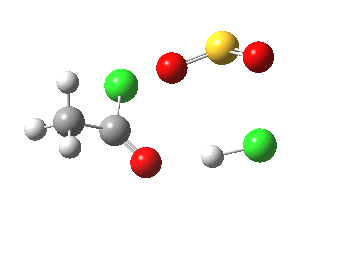
- The initial feature (IRC -2 to 0.0) is the cleavage of the C-O bond (1.862Å at the transition state)
- This point is 28.7 kcal higher in ΔG than the initial reactants, and is the highest energy point in the mechanism. As noted earlier, additional solvation-stabilisation involving discrete hydrogen bonds from e.g. acetic acid, is likely to lower this energy.
- This is followed (IRC +1.0 to +2.0) by a proton transfer from oxygen to chlorine.
‡ I have discussed elsewhere the conventions used in arrow pushing. Nick uses the “American system” , whereas in this blog, I use a system I will call the Charles Rees method. I prefer this one, since it nicely maps onto more elaborate ways of identifying electron pairs in molecules, such as ELF and QTAIM, which themselves are based on quantum mechanics. Nick’s system differs mostly in the end-point for the arrows which he directs towards atoms whereas I direct them towards bonds. It might also be an interesting discussion point as to what criteria should be used to define three-dimensional arrow pushing; in effect the path that the arrow takes and what (pedagogic) meaning this might have.
₪ Following the initial proton transfer from Cl to oxygen, a very shallow minimum ion-pair is formed as a prelude to forming the C-Cl bond in a second step. This is because the additional oxygen present in a carboxylic acid stabilises the intermediate oxenium cation.
My “intuition” would have predicted initial addition of acetic acid to the S=O double bond (not the S–Cl single bond), generating MeCO2-S(OH)Cl2 as intermediate, followed by HCl expulsion to chlorosulfit anhydride, and then the re-addition of HCl to C=O.
This is because “side-on” additions should be stereoelectronically easier to pi rather than sigma bonds. Could this bring down the energy of the initial step, at the cost of an additional intermediate?
Ah, but another factor which is actually very important in such mechanisms is the ability of the TS to adopt something like a linear orientation of any H-bond or H-transfer. Addition across the S=O bond (in the required six membered ring) results in a very bent O…H…O=C angle. I will try to calculate this option to see if it indeed has a higher (or a lower) energy.
…. 18 hours later:
The free energy of this transition state relative to the starting material is 32.5 kcal/mol, higher than the previous transition states. Because co-linear alignment of the O-H…O was not possible, the proton transfer did not happen. Instead, the reaction corresponds to a Walden-like inversion at S to generate an ion-pair:
Notice how the chlorine atom displaced by the incoming oxygen nucleophile emerges anti, but then rapidly rotates around to minimise the resulting ion-pair dipole energy. One might imagine that this chlorine anion now wanders over to the O-H bond to abstract the proton, and the mechanism then continues as shown in the post.
[…] The mechanism (in 4D) of the reaction between thionyl chloride and a carboxylic acid. […]