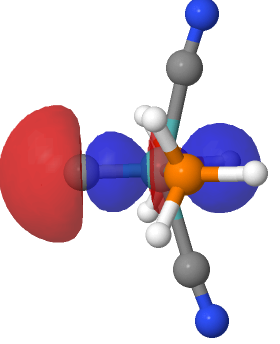
Introductory chemistry will tell us that a triple bond between say two carbon atoms comprises just one bond of σ-axial symmetry and two of π-symmetry. Increasingly mentioned nowadays is the possibility of a quadruple bond between carbon and either itself or a transition metal, as discussed in the previous post. Such a bond comprises TWO bonds of σ-axial symmetry. Since most people are unfamiliar with such double bonds and in particular with how that second σ-bond sits with the first, I thought it would be interesting to show such an orbital. This one is a localised orbital 41, selected from the previous post for the molecule (PH3)2(CN)2Mo⩸C.
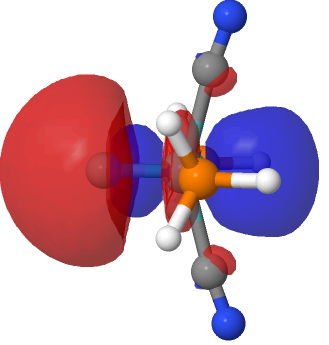
| NBO 41, threshold 0.040 au | NBO 41, threshold 0.018 |
|---|---|
 |
 |
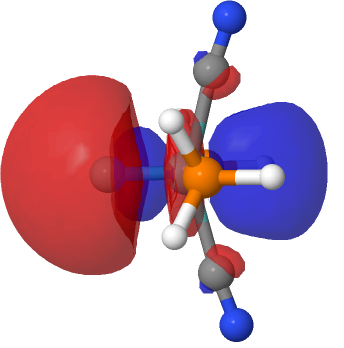
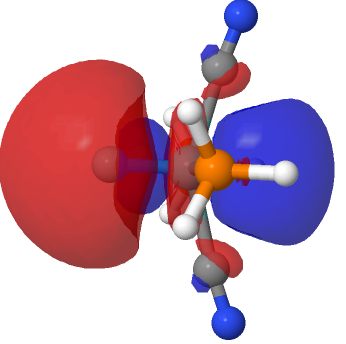
| NBO 41, threshold 0.016 | NBO 41, threshold 0.014 |
 |
 |
| NBO 41, threshold 0.012 | NBO 41, threshold 0.007 |
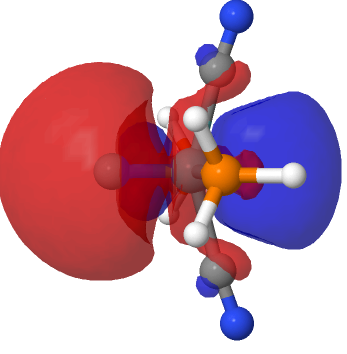 |
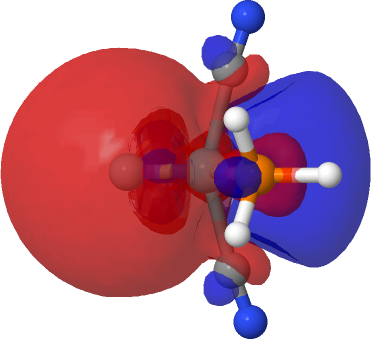 |
The above shows how the orbital changes with the isosurface threshold. At high values, it looks very similar to the normal σ-bond but as the threshold gradually decreases, a second “sheath” starts to surround the inner orbital until the latter is entirely enclosed. This orbital has a node not so much along the bond itself, but between the inner and outer layers of the bond, which is how the two σ-bonds are differentiated. This effect was first noted in 2016 in terms of the compound CH3F2-, in which an expanded carbon valence shell creates a second σ-bond.
Certainly not a representation that has ever appeared in a text book I think! But perhaps one that chemists may have increasingly to become familiar with.
Appendix Here are some superimposed orbitals to facilitate comparisons. Firstly orbital 41 (the higher energy σ-orbital) with orbital 22 (the lower energy σ-orbital). The first has yellow/green for the two phases, the second has red/blue.
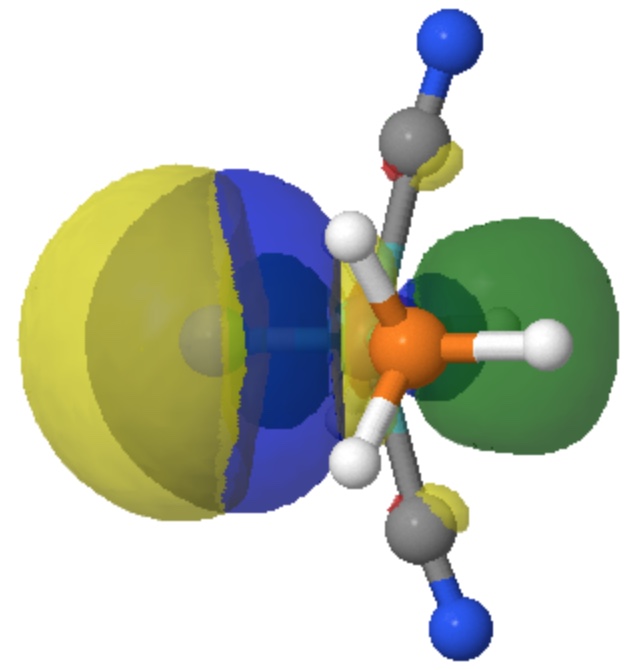
Next, σ-orbital 22 (yellow/green) with orbital 42 (red/blue) surrounding it, revealing the avoided overlaps (Pauli repulsions) between the two by virtue of having orbital 42 unoccupied.
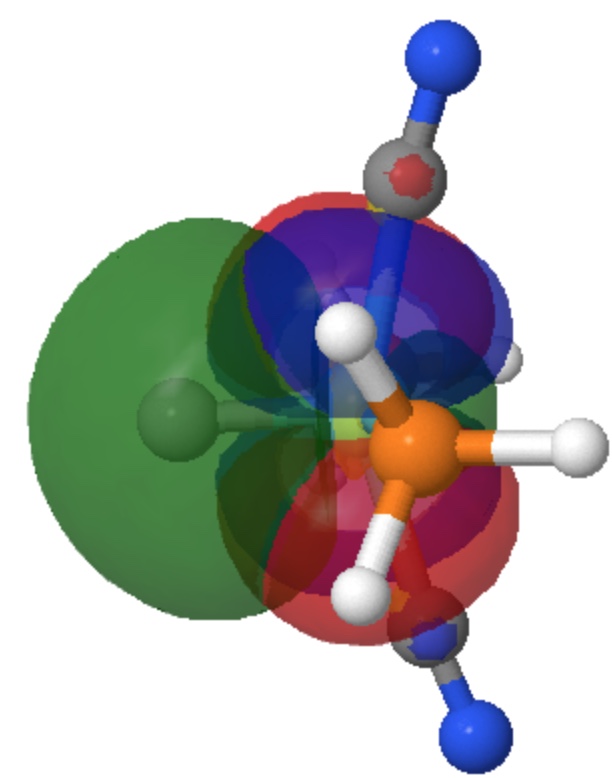
Next, σ-orbital 41 (yellow/green) with orbital 42 (red/blue) surrounding it, revealing the reduced overlap between these two.
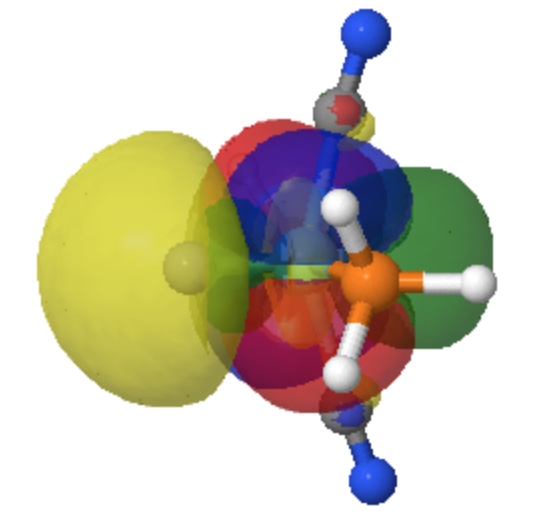
Appendix 2 A “pure” form of the double-layered σ-bond can be seen with the diatomic molecule Ti2, contoured at 0.0225 au. The red phase is about to join in the middle.
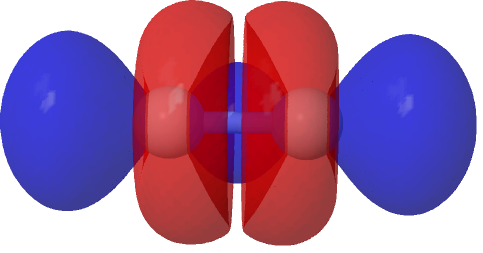
The electron density from this orbital is shown below and shows clearly the two layers of density comprising the σ-bond, with the outer layer at this isosurface value (0.00052 au) about to join up in the middle to complete the outer sheath. I have left it unjoined so that you can see “inside layer”, since translucency does not always get the message across.
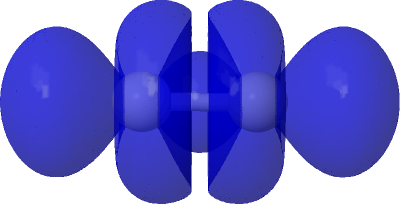
This is NBO 41 for Mo and NBO 40 for Os. In both cases the other sigma bond is NBO 22 – it would be interesting to see that plotted in the same way and to the same scale, to get an idea of the interactions between the two.
I will superimpose the two orbitals using JSmol. That would do what you want.
Thankyou for that appendix, it is a very useful presentation.