In 2010 I recounted the story of an organic chemistry tutorial, in which I asked the students the question “how would you synthesize 3-nitrobenzonitrile“.
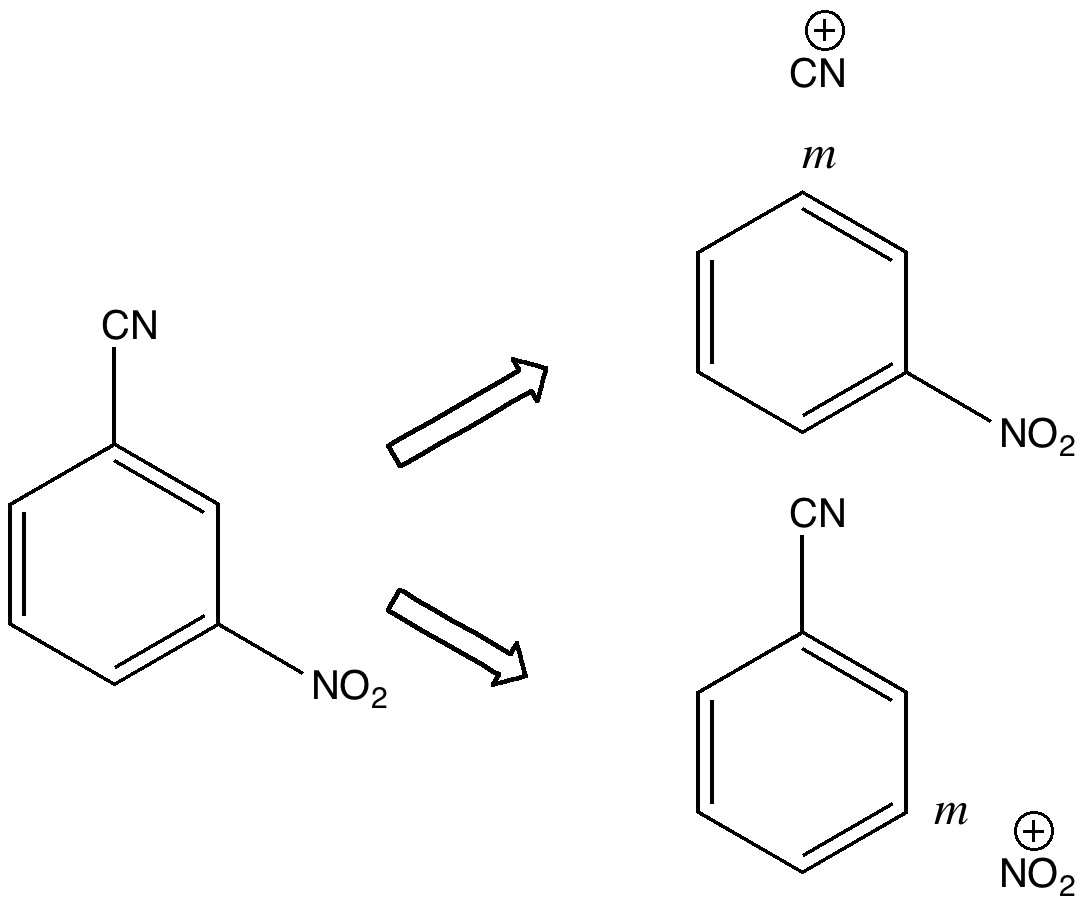
The expected answer was to generate a nitronium ion to nitrate benzonitrile, but can one invert this by generating a C⩸N+ ion to cyanate nitrobenzene? The students were then invited to generate a valence bond structure for C⩸N+ and I showed them the possibility that it might contain a quadruple bond to the carbon. Ten years later, Mike Turner in a comment on that post revealed an article dating from 1960[1] in which cyanogen fluoride was studied. There, in an innocuous comment, they state “Cyanogen fluoride, like cyanogen chloride, re-acted with benzene in the presence of aluminum chloride to form benzonitrile in 20% conversion“. Here I explore whether this reagent really can be a source of free C⩸N+.
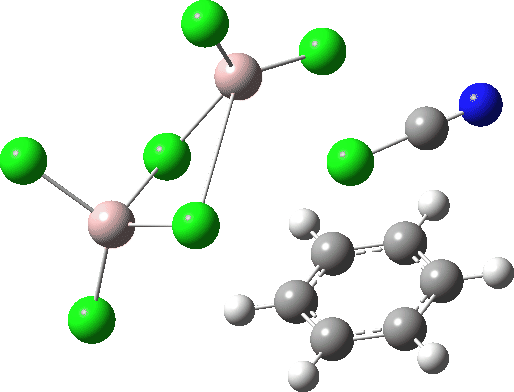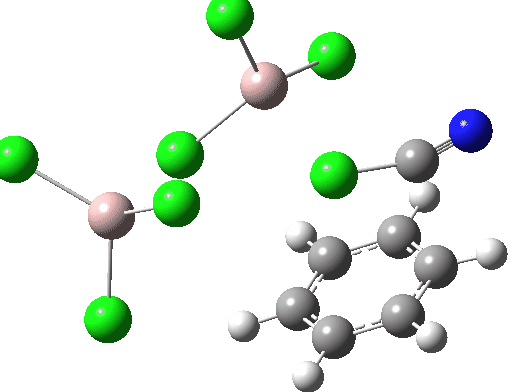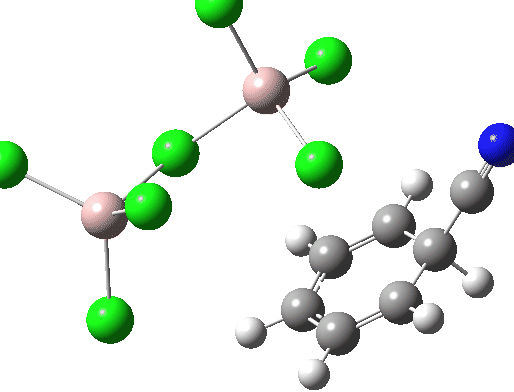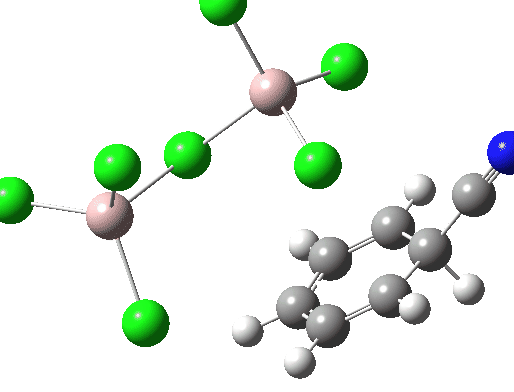
Calculations at the ωB97XD/Def2-TZVPP/SCRF=water level (FAIR Data DOI: 10.14469/hpc/7584) were conducted to explore the possible energetics of using ClCN to electrophilically cyanate benzene. Firstly, the energy of the separated ion pair AlCl4–.C⩸N+ is 190.9 kcal/mol higher than the neutral reagents AlCl3.ClCN, which makes the formation of free C⩸N+ unlikely. So what about a concerted process, in which benzene as a nucleophile attacks ClCN with the help of AlCl3? This would be a 1,1-substitution reaction at an sp-carbon centre. 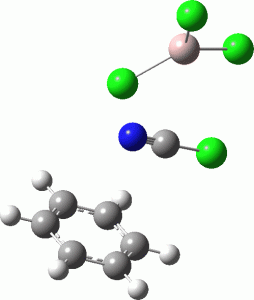
The free energy barrier for this bimolecular process is 46.4 kcal/mol. It is along the right lines, but still about 20 kcal/mol too high for a facile thermal process.
In an effort to improve the model and hence reduce this barrier, the dimeric reagent Al2Cl6 was tried instead. The final free energy barrier was 48.5 kcal/mol.
So to conclude, generating free C⩸N+ is very unlikely. But cyanogen chloride can act as a C⩸N+ “delivery agent” via a bimolecular route. There remains a mystery however. The free energy barriers for our two models are too high to accomplish facile cyanation of benzene. There must be another mechanism, as of yet unexplored, which must be found to finish off this study!
References
- F.S. Fawcett, and R.D. Lipscomb, "CYANOGEN FLUORIDE", Journal of the American Chemical Society, vol. 82, pp. 1509-1510, 1960. https://doi.org/10.1021/ja01491a064
How about a multistep reaction in which first a solvent replaces Cl- then the Sol-CN+ reacts. Have you tested this path?
Unfortunately from the simple statement found in the article, we do not know how the experiment was conducted, or the solvent used. Assuming e.g. benzene as both reagent and solvent solves the problem that you pose. I used water as a model solvent to try to mimic the possible polarity of a solution of aluminium chloride in benzene. A lesser dielectric would increase the barrier. Note that at the transition state, the dipole moment is only ~9D, which is not particularly polar.
The reaction with ClCN was already studied by Friedel and Crafts (Ann. Chim. 1884, p. 528; available via Gallica.bnf.fr) who got low yields. Two preparative papers by Paul Karrer in Helvetica (10.1002/hlca.19190020150 and 10.1002/hlca.19200030126) give procedures for the reaction with BrCN or ClCN and arenes (benzene, toluene etc.), either neat or in CS2 as solvent to provide nitriles in high yields.
From the experiment description, one uses a suspension of AlCl3 in arene (with or without CS2), adds BrCN (or introduces ClCN gas). The reaction starts spontaneously or after heating, with evolution of HCl/HBr. Sometimes standing at r.t. is sufficient.
In my opinion one might want to check if intermediary addition of HCl to X-CN could provide a more reactive reagent such as Cl2C=NH, or alternatively if protonation to [X-CN-H]+[Al2Cl7]– is an option.
At least from reading Karrers descriptions, it might be that an induction period could apply, in which a little HCl is generated. Later, the latter would be more abundant, as it is a reaction product.
Would it improve the electrophilic strength of the carbon if another aluminium coordinated to the nitrogen? Not good for the entropy factor though.
The suggestion to use an additional Al to coordinate to N, along with Lukas’ suggestion of HCl protonation which is related, will be followed up in the next post.
Dicationic electrophiles seem likely, see https://doi.org/10.1021/ja00116a009.