NCI (non-covalent interactions) is the name of a fascinating new technique for identifying exactly these. Published recently by Johnson, Keinan, Mori-Snchez, Contreras-Garca, Cohen and Yang, it came to my attention at a conference to celebrate the 20th birthday of ELF when Julia Contreras-Garcia talked about the procedure. It is one of those methods which may seem as if it merely teases out the obvious about a molecule, but it is surprising how difficult seeing the obvious can be sometimes. I have blogged about this previously, in discussing the so-called Pirkle reagent. On that occasion, I used the QTAIM technique to identify so-called critical points in the electron density. NCI goes one stage further in identifying surfaces of interaction rather than just single points, the idea being that this focuses attention on regions in molecules which are primarily responsible for binding, stereoselection and other aspects of molecular selectivity.

The Pirkle reagent
So I was intrigued as to whether the NCI method might find something that my analysis using the QTAIM procedure might have missed. The required program is available for download. I will not go into the theory behind the program, but like AIM, it uses the properties of the electron density via a combination of the first and second derivatives to concentrate on the non-bonded or weakly interacting regions of a molecule.
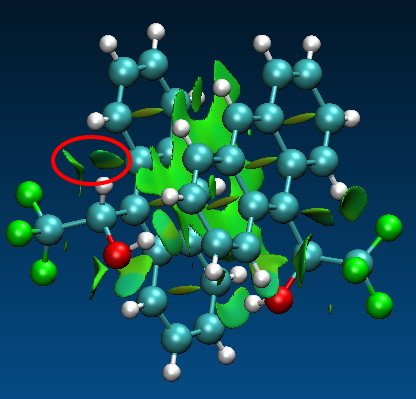
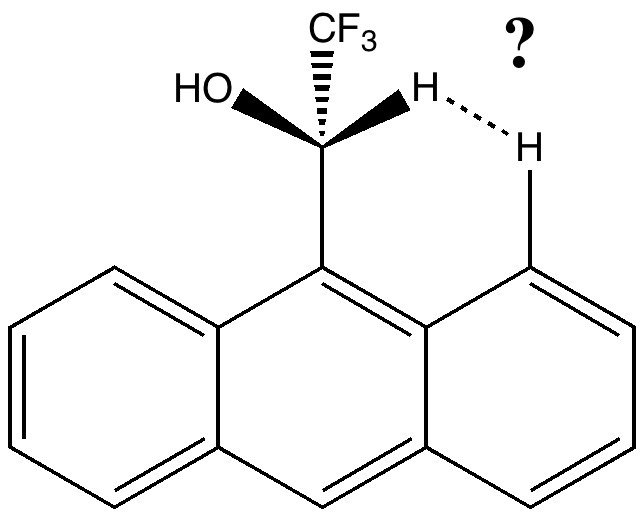
The results of the analysis (using the SCF option in the program, and a B3LYP/6-31G calculation) are displayed using VMD, and I cannot pull my usual trick of displaying the surface within the page of a blog via Jmol (although it seems Jmol with some effort could probably be persuaded to also render the information). So the above cannot be rotated. I have therefore circled one (there are others) interesting region in red. This encloses two surfaces. I should explain the colour coding adopted by the program. Red would be a repulsive interaction, and blue attractive. Weak interactions are shown in green. In the diagram above, these include the π-π stacking and various hydrogen bonds. But concentrating on the two surfaces inside the red circle, one occurs between the two hydrogens shown below. It catches the eye because there is a blue-tinge to the colour coding! This might mean it is a bit stronger than just “weak”.
The NCI method I do not think is meant to provide a definitive answer to the question; is that interaction real/strong? It serves, as I noted earlier, to spike interest. Here, it does that, since this particular interaction had indeed never previously been identified for attention (the obvious had been missed!). Highlighting such potential regions of a molecule and perhaps then helping in the design of experiments to test if the interactions are real is what the NCI program is meant to do (IMHO)!
Tags: Interesting chemistry, Julia Contreras-Garcia, watoc11
[…] how one might proceed to analyse the origins of the stereoinduction, the NCI interaction surfaces (as described in this post) are shown below. Note how the extensive degree of green interaction surface is associated with the […]
[…] The previous post set out a problem in conformational analysis. Here is my take, which includes an NCI (non-covalent interaction) display as discussed in another post. […]
[…] a second one missed the first time round (labelled 2 in that post). Then a method known as NCI was tried, which revealed an H…H interaction, labelled ? in that post! Here I discuss the origins of […]