In the previous post, I showed that carbon can act as a hydrogen bond acceptor (of a proton) to form strong hydrogen bond complexes. Which brings me to a conceptual connection: can singlet dicarbon form such a hydrogen bond?
Dicarbon can be variously represented as above. The first form shows it as a bis-carbene, with an unbonded lone pair of electrons at each end of a carbon double bond. The middle form has emerged in the last ten years or so as a serious alternative to describing the singlet state structure. It contains a so-called triple endo–bond and one further much weaker exo-bond (indicated separately by the symbol above the bond), referred to for simplicity as quadruple-bonded dicarbon. The third form would be a triplet biradical with triple bonded carbon. The species is known to be a singlet ground state with a significant excitation energy to the triplet.‡ One can then ask the question: would either of these singlet state species be capable of being a hydrogen bond acceptor?
Time for calculations, at the CCSD(T)/Def2-TZVPP level using HF as the hydrogen bond donor (to enable advantage to be taken of the axial symmetry), data DOI: 10.14469/hpc/6554.
- The singlet quadruple bonded form emerges as 32 kcal/mol higher in total energy than the singlet dicarbene.
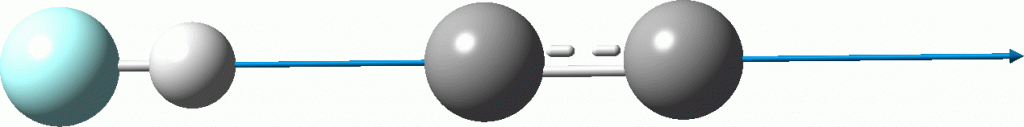
- The quadruple bonded form shows no sign of forming a hydrogen bond. The geometry optimisation curve is shown below followed by the final geometry (Å).
- The bis-carbene form (calculated by a double electron excitation, orbitals 10 to 12 and 11 to 15) DOES form such a complex. The hydrogen bond length (2.04Å) is exactly that found from the crystal structures of the shortest such bonds.
- Two of the normal vibrational modes of this species are shown below, being respectively the H…C and C=C stretches (153 and 1394 cm-1).

So dicarbon CAN form a short hydrogen bond to a donor such as HF, but only in its excited singlet state, which is some 32 kcal/mol above the quadruple-bonded form. Perhaps because of that fourth bond, the hydrogen bonding ability of this species is entirely inhibited. We have gotten to the point I wanted to reach; an experimental prediction that if singlet dicarbon can ever be trapped in a very inert matrix at very low temperatures in the presence of a hydrogen bond donor, it will not form a hydrogen bond to that donor. That is going to be a difficult experiment, but at least the prediction is out there as a challenge!
‡At the same level of theory, the triplet 3Σu state is calculated to be 27.0 kcal/mol higher than the singlet 1Σg.

