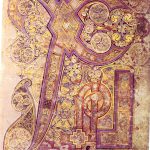
The Book of Kells is a spectacularly illuminated gospel manuscript dating from around 800AD and held in Trinity College library in Dublin. Some idea of the colours achieved can be seen below.
I thought it would be of interest to list how these colours were achieved.
- Black ink was made from oak-galls mixed with iron sulfate and acetic acid from wine or vinegar. Carbon-black ink was used less frequently
- Greens made by mixing arsenic sulfide and indigo (extracted from the Woad plant) or using verdigris, which is a copper carbonate or chloride.
- Red dots were made from red lead, or Pb3O4.
- Blue used indigo, not lapis lazuli.
- White was gypsum or calcium sulfate.
- Yellow was arsenic sulfide, known as auripigmentum since it had a lustrous golden quality.
- Purple came from a lichen (Roccella tinctoria) which is better known to chemists as a source of litmus; the basic chromophore of which is 7-hydroxyphenoxazone.
If you ever visit Dublin, do go and see the manuscript for yourself.
Tags: Biblical manuscripts, Book design, Book of Kells, Books, City: Dublin, County Meath, Dublin, Hospitality/Recreation, Illuminated manuscript, Kells, Manuscripts, Trinity College, Trinity College Dublin, Trinity College Library, Trinity College Library Dublin, Western art, Woad plant