One future vision for chemistry over the next 20 years or so is the concept of having machines into which one dials a molecule, and as if by magic, the required specimen is ejected some time later. This is in some ways an extrapolation of the existing peptide and nucleotide synthesizer technologies and sciences. A pretty significant extrapolation, suitable no doubt for a grand future challenge in chemistry (although the concept of tumbling a defined collection of atoms in a computer model and seeing what interesting molecules emerge, dubbed with some sense of humour as mindless chemistry, is already being done; see DOI: 10.1021/jp057107z).
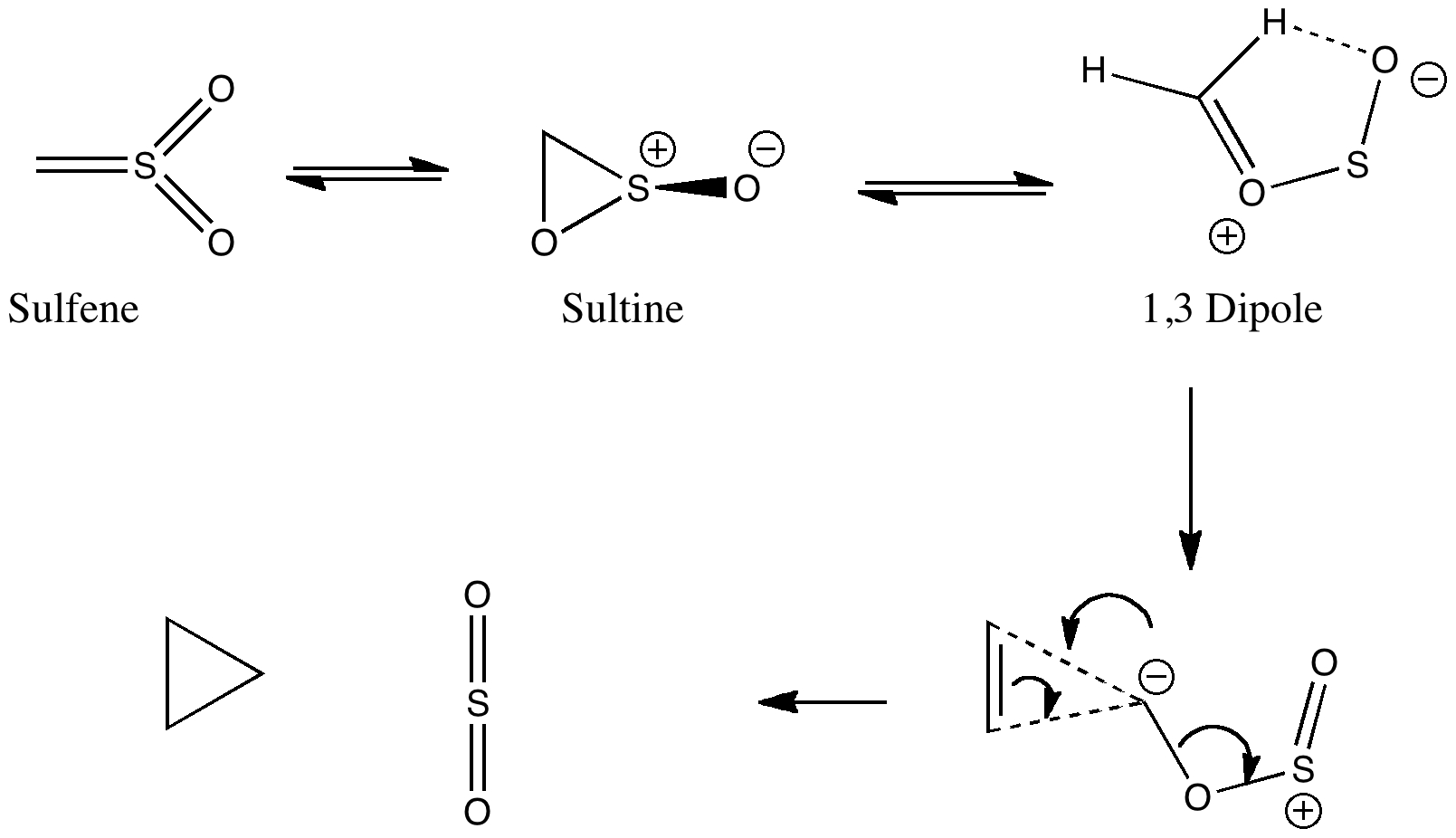
Well, let us return to present day reality (I know it was a little unfair to capture your attention with such a grand title!). Consider the sequence above. Sulfenes are known simple elaborations of sulfur trioxide, with one oxygen replaced by a CH2 group. They can exist as isomeric rings, known as sultines (and which are of similar energy to the sulfenes, see DOI: 10.1016/j.theochem.2007.10.035). Few people have speculated upon what might be done with this small collection of atoms. It struck me (I am unaware it has struck anyone else, but I am happy to be corrected) that it might be useful as a reagent for delivering a carbene. The precedent is that oxaziridines (in which the SO unit is replaced by e.g. NR) can be used to transfer either oxygen or NR to alkenes, and dioxiranes (in which the SO unit is replaced by an oxygen) are very useful reagents for oxygen transfer to an alkene. In the example of the sultine, loss instead of carbene (CH2) would result in the thermodynamically stable sulfur dioxide. Also apparent is that the sultine is asymmetric (chiral) and so perhaps there is also a prospect of delivering that carbene asymmetrically (a reaction normally done with the help of metal catalysts). As shown above, the carbene is also nucleophilic, rather than electrophilic, which may also be useful in some contexts.
Enter the computer, which will be used to see if these simple ideas can be turned into the design of a new reaction. Firstly, the assertion that the reaction producing cyclopropane and sulfur dioxide is exothermic is easily tested (B3LYP/cc-pVTZ); it comes out as exothermic in free energy by -26.6 kcal/mol (some of which of course is due to entropy). Next, the transition state for the delivery.

Transition state for carbene transfer from sulfine
This emerges (DOI: 10042/to-4476) with a free energy barrier of 37.4 kcal/mol relative to the sultine. Rather too high a barrier to constitute a useful synthetic reaction! But there is something interesting to be learnt from this transition state. Whilst the product is clearly cyclopropane and sulfur dioxide, the reactant is not the sultine but appears to be another species, labelled above as the 1,3 dipole (DOI: 10042/to-4487), a species which is 13 kcal/mol higher in free energy than the sultine itself (but does it have to be formed first, or is it merely on the reaction path?). There are other noteworthy aspects of the transition state. The carbene cycloaddition is a 4n electron process, with an apparent antarafacial component, this mapping onto inversion at the carbene centre. The bond formation at the alkene is very asynchronous, and the SO2 unit clearly does appear to act as a chiral auxilliary. Also these aspects would have to be factored into the eventual design.
We now enter an optimization stage of the process, in which we try to reduce the activation barrier in order to produce a viable reaction. Replacing CH2 by CF2 however increases the barrier to 42.5 kcal/mol, whilst substituting Se for S induces a barrier of 40.6 kcal/mol. More variation of the various substituents (including the alkene) will be needed to see if such a reaction could actually be carried out, but this is relatively routine process, not attempted here (perhaps not entirely routine; thus predicting what might happen is easy compared to analyzing what does not happen, see DOI: 10.1002/anie.200801206). So, there is certainly no claim here that a new reaction has been designed. Rather a tentative hint at the kind of processes that might be involved, eventually, in dialing a molecule.
Tags: free energy, free energy barrier, Interesting chemistry, metal catalysts, nucleotide synthesizer technologies, similar energy