I am now inverting the previous question by asking what is the largest angle subtended at a chain of three connected 4-coordinate carbon atoms? Let’s see if further interesting chemistry can be unearthed.
Specifying only angles > 130°, the following distribution is obtained.
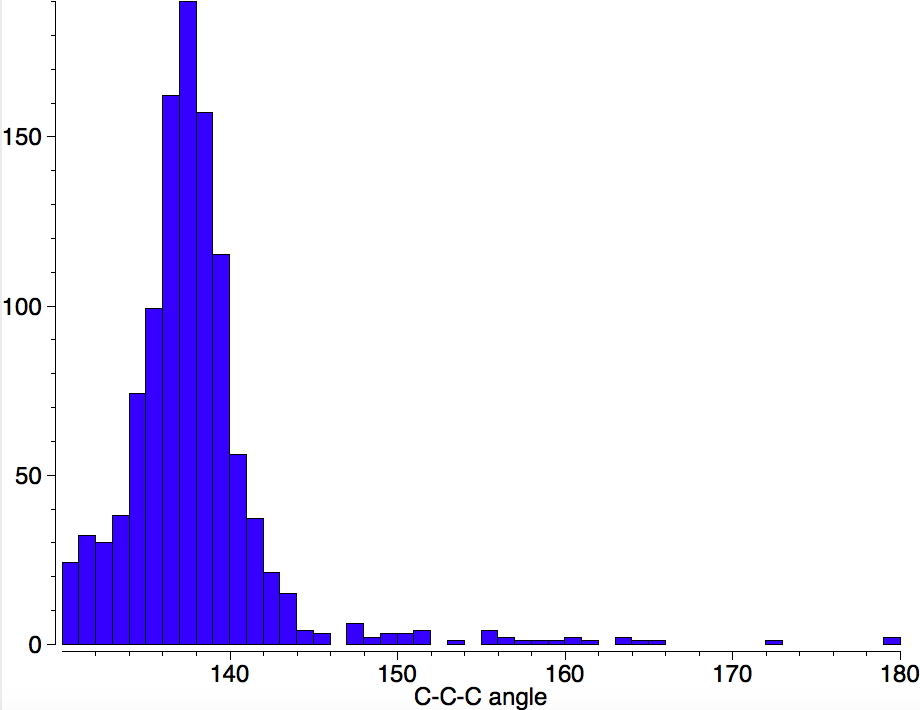
- Note the maximum at ~138°. This is typical of that found in spiro-cyclopropanes, although I have not checked if other kinds of compound can also sustain this angle.
- There appear to be a few examples at 180° but these appear to be simple errors in the crystal coordinates.
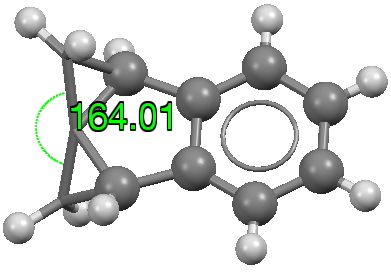
- The first real example occurs at 166°[1] and contains an almost hemispherical carbon atom, doi: 10.5517/CCZBB2P [2]

- A second example is a sprung spiro-cyclopropane [3] in which the large angle is maintained without the help of a metal.
- This latter example suggests that a structural modification as shown below might take the angle to almost 180° (calc. ωB97XD/Def2-TZVPP = 177.2°[4]).
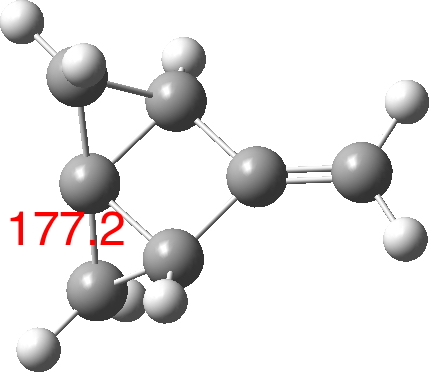
It is remarkable how much the standard angle subtended at four-coordinate carbon (109.47°) can be opened. It makes one wonder whether something approaching 180° is achievable, and what the properties of such a molecule might be.
References
- V. González-López, M.A. Leyva, and M.J. Rosales-Hoz, "Coupling of acetylene molecules on ruthenium clusters, involving cleavage of C–Si bonds in the alkyne and coordination of a phenyl ring of a SiPh3 group", Dalton Transactions, vol. 42, pp. 5401, 2013. https://doi.org/10.1039/c3dt32335h
- Gonzalez-Lopez, V.., Leyva, M.A.., and Rosales-Hoz, M.J.., "CCDC 903652: Experimental Crystal Structure Determination", 2013. https://doi.org/10.5517/cczbb2p
- R. Boese, D. Blaeser, K. Gomann, and U.H. Brinker, "Spiropentane as a tensile spring", Journal of the American Chemical Society, vol. 111, pp. 1501-1503, 1989. https://doi.org/10.1021/ja00186a058
- H. Rzepa, "VAJHAP", 2016. https://doi.org/10.14469/hpc/1861
Tags: Angle, metal, Subtended angle
Roald Hoffmann has sent me the following comment:
“On large angles: Computationally, we found some in hypothetical prismane structures, attached (doi: 10.1021/jo034910l and 10.1021/om960099b) We also did an estimate of the energy to open up that angle in substituted methane, in another paper. The structure your search found are intriguing.