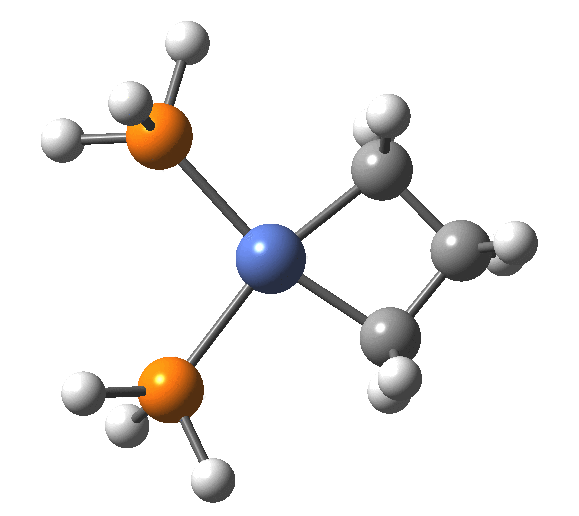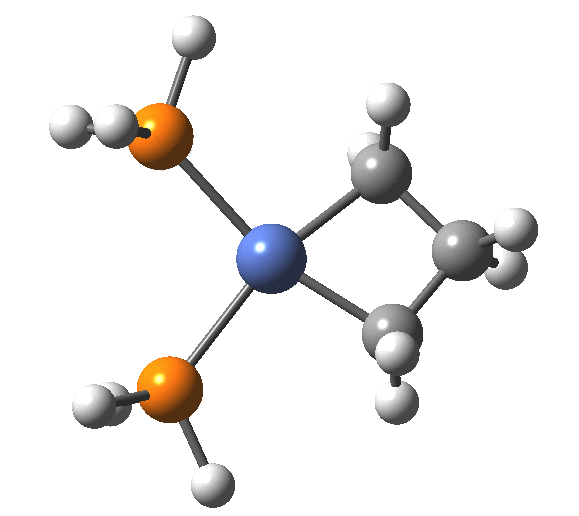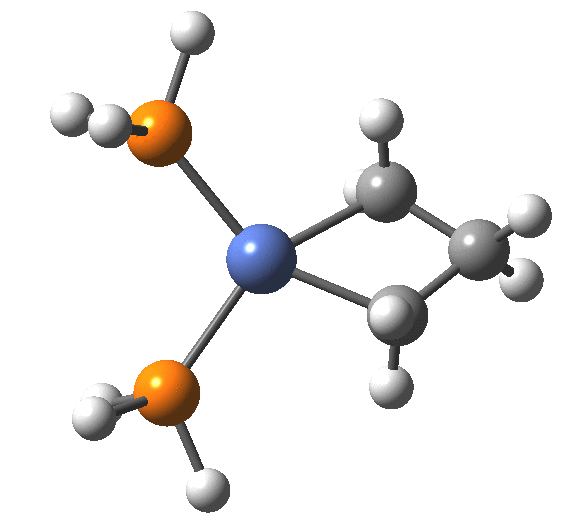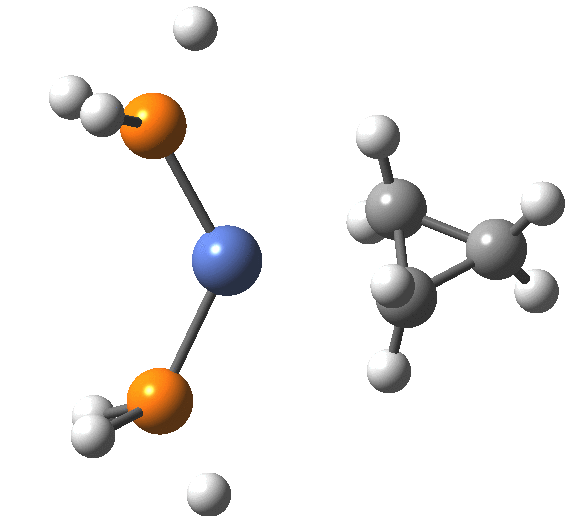
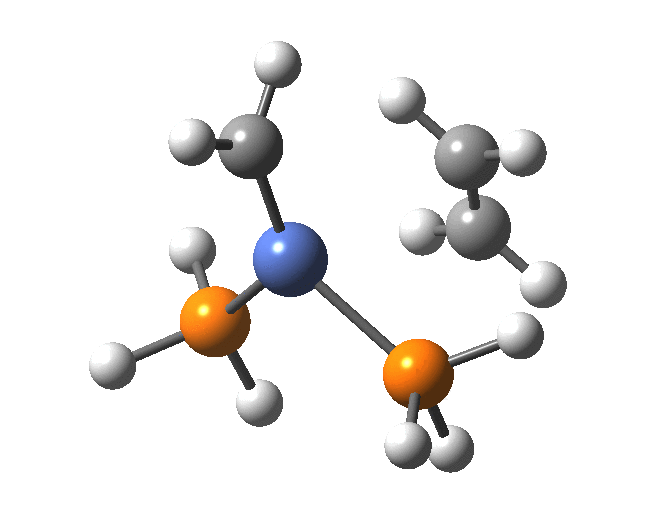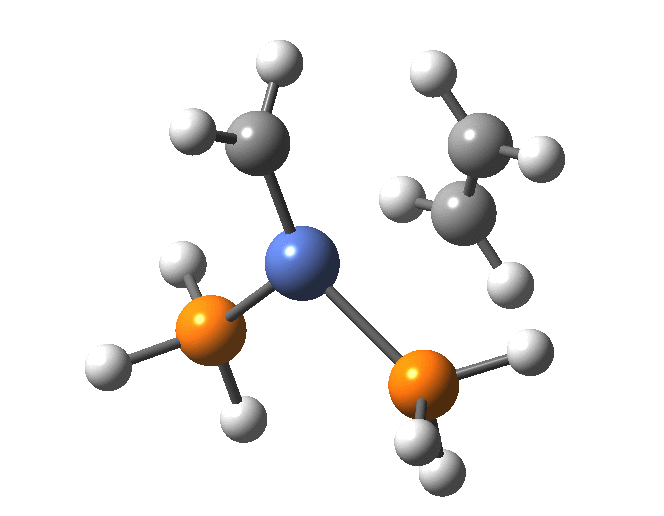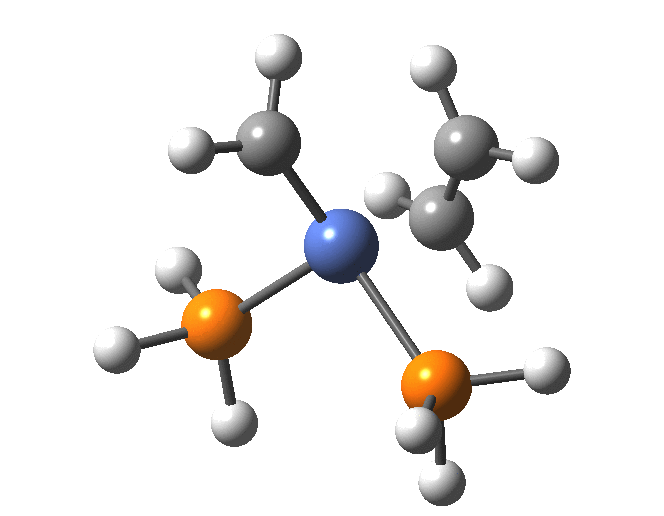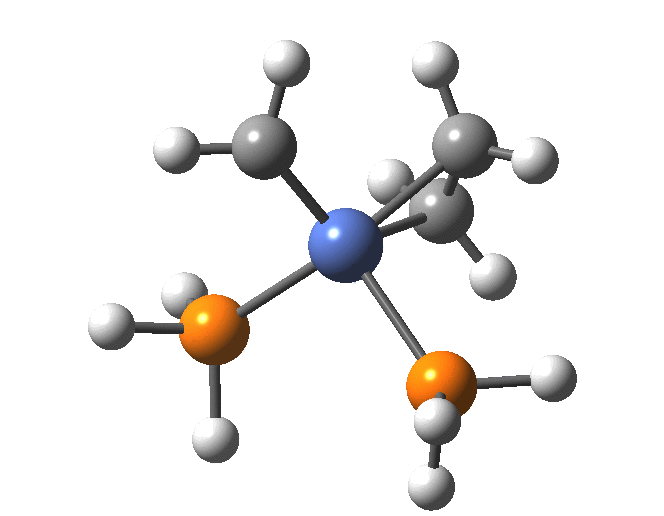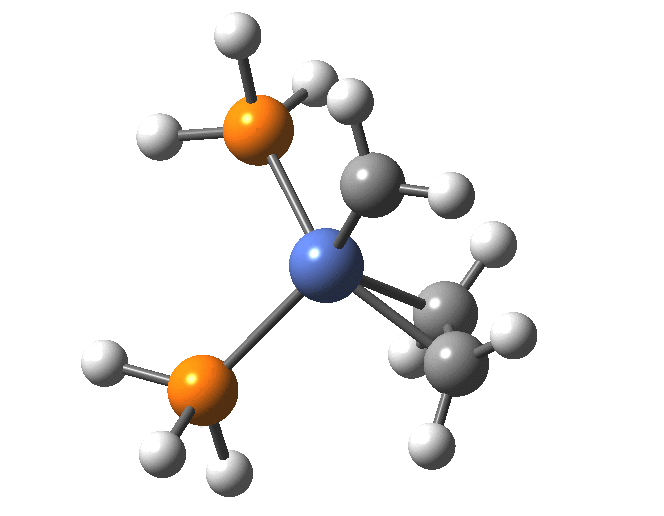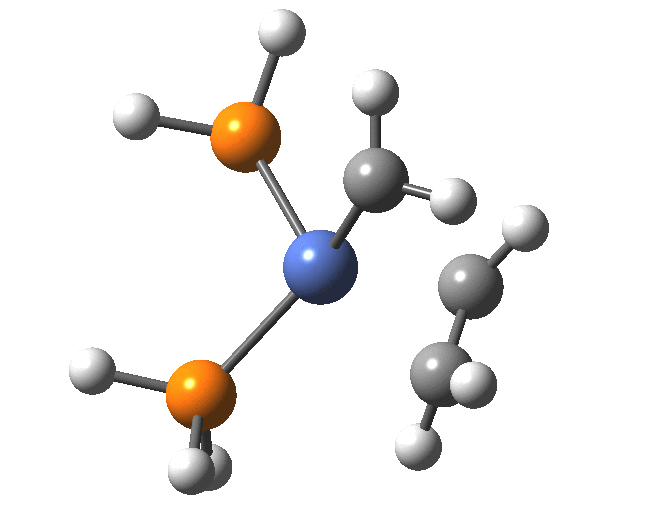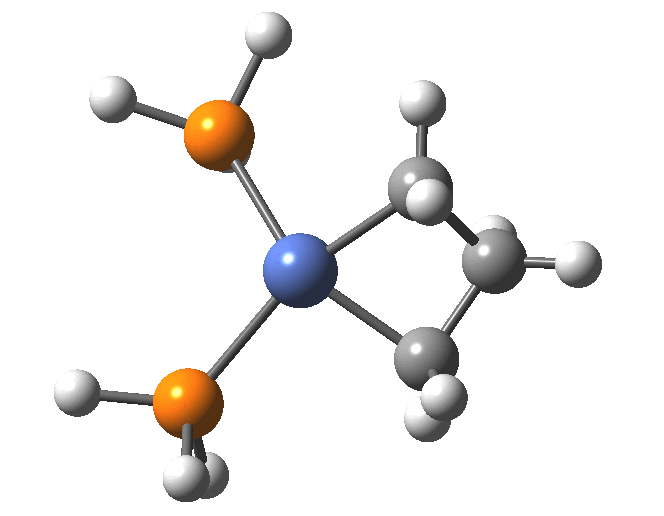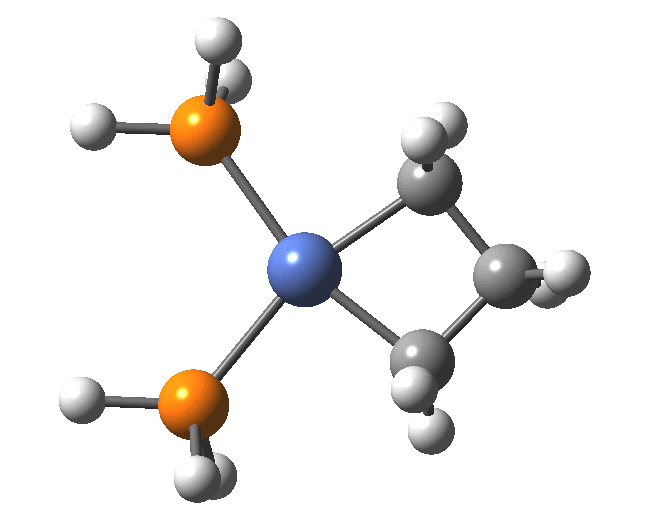A fascinating re-examination has appeared[1] of a reaction first published[2] in 1960 by Wittig and then[3] repudiated by him in 1964 since it could not be replicated by a later student. According to the new work, the secret to a successful replication seems to be the presence of traces of a nickel catalyst (originally coming from e.g. a nickel spatula?). In this recent article[1] a mechanism for the catalytic cycle is proposed. Here I thought I might explore this mechanism using calculations to see if any further insights might emerge.
In the mechanism above (I have retained the original numbering shown in the article itself), Ln is set to 2PH3 as an initial approximation and the solvent thf is approximated only by a continuum solvation field, with no explicit thf molecules involved at this stage. At this level and using ωB97XD/Def2-SVPD/SCRF=thf free energies, one can explore the cycle quite quickly (~2-3 days). It is also interesting that this reaction unusually involved nine different elements (I wonder what the record is? Not much greater I suspect).
| Species | ΔΔG298 | DataDOI |
|---|---|---|
| 4+CH2NMe3+LiOTf + ethene | +23.9 | [4],[5] |
| 5 | 0.0 | [6] |
| TS (5→ 9) | 12.7 | [7],[8] |
| 9 + LiOTf + NMe3 | 0.2 | [9] |
| TS (9 + ethene → 6) | 7.2 | [10],[11] |
| 6 | 4.8 | [12] |
| TS (6 → 7) | 11.2 | [13],[14] |
| 7 | -36.3 | [15] |
| TS (7 → 4+8) | -18.8 | [16],[17] |
| 4+8 + LiOTf + NMe3 | -29.7 | [4] |
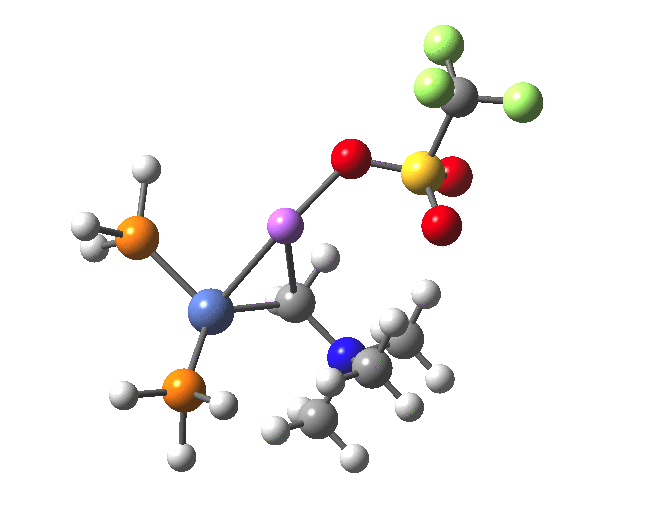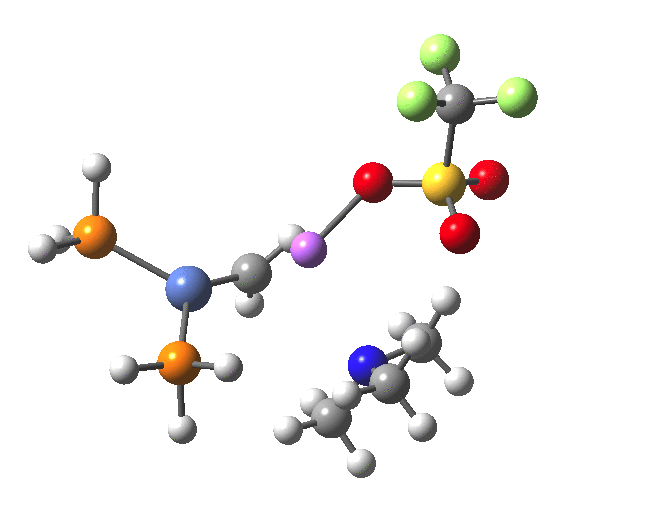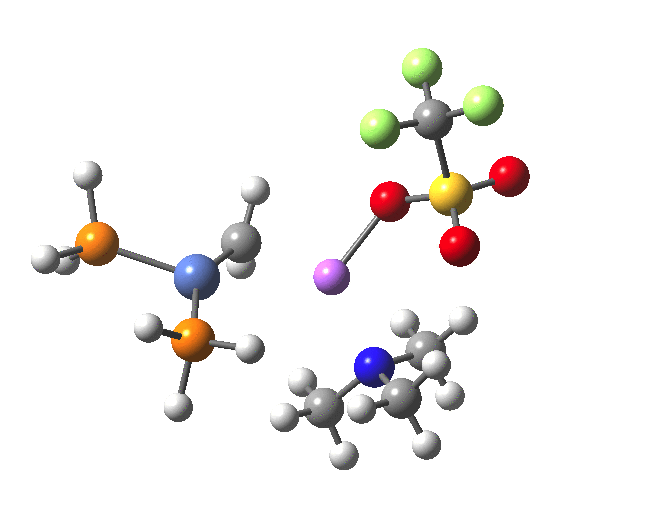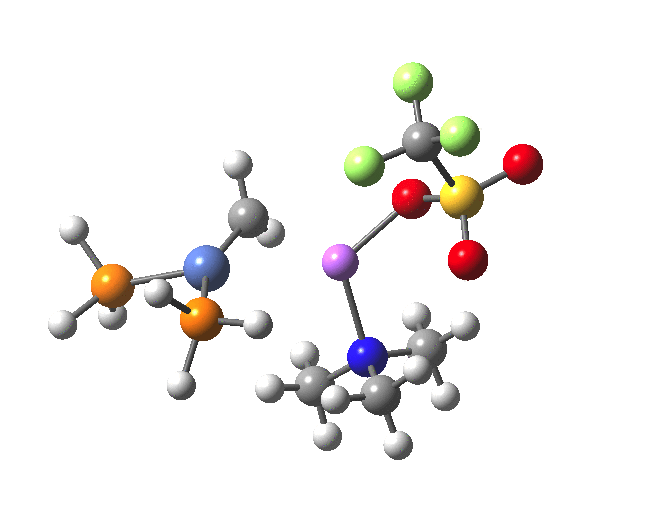
The structure of the complex 5 is more or less as shown in the article. The mean single bonded Ni-C length in the Cambridge structure database (CSD) is ~1.9Å, and (formally at least) Ni=C lengths are shorter at ~1.80-1.85. There is one reasonable analogy to the sub-structure shown below[18],[19] with a C-Ni length of 1.90, Ni-Li = 2.51 and Li-C = 2.40 which is reasonably similar to what is shown below.
T

Click for 3D
The elimination of NMe3 reveals a reasonable thermal barrier, resulting in the formation of the nickel-carbene product and the complex between NMe3 and LiOTf.
The Ni-carbene then reacts with alkene (modelled here by ethene) to form a Ni-alkene π-complex, with a very low barrier to the exo-energic reaction.
This complex then rearranges, again with a small barrier, to the metallocyclobutane, with considerable release of energy.
Finally, the metallocyclobutane extrudes the nickel to form cyclopropane bound to the Ni(PH3)2 as a pseudo-π/agostic complex, with this step of the reaction being somewhat endo-energic (+6.6 kcal/mol). As modelled, it produces a low-coordination Ni product 4, which also causes the initial reactants to be relatively high in energy (+23.9 relative to 5). This suggests that the entire cycle should optimally be repeated by including say two explicit thf solvent molecules, which could coordinate to 4, thus lowering its energy relative to the rest of the cycle.
Below is shown the NCI (non-covalent-interactions) surface for the Ni-cyclopropane complex, revealing the relatively high density between the Ni and the edge of the cyclopropane (high enough indeed to be considered on the verge of being covalent density). No examples of this motif are found in the CSD.

Click for 3D
Overall, the reaction as shown shows entirely reasonable energetics and activation free energy barriers (with the caveat that inclusion of explicit solvent molecules might improve things, see above). We might conclude from this that the catalytic cycle as proposed is entirely reasonable. What we cannot comment on of course is the relative energetics of any of the competing side reaction shown in the original scheme,[1] but it would be really easy to include them in a more complete analysis if needed. I wanted to show here that a simple reality check on a proposed reaction mechanism can be quick to perform, and perhaps nowadays should be regarded as a sine qua non of mechanistic speculation.
References
- S.A. Künzi, J.M. Sarria Toro, T. den Hartog, and P. Chen, "Nickel‐Catalyzed Cyclopropanation with NMe<sub>4</sub>OTf and <i>n</i>BuLi", Angewandte Chemie International Edition, vol. 54, pp. 10670-10674, 2015. https://doi.org/10.1002/anie.201505482
- V. Franzen, and G. Wittig, "Trimethylammonium‐methylid als Methylen‐Donator", Angewandte Chemie, vol. 72, pp. 417-417, 1960. https://doi.org/10.1002/ange.19600721210
- G. Wittig, and D. Krauss, "Cyclopropanierungen bei Einwirkung von <i>N</i>‐Yliden auf Olefine", Justus Liebigs Annalen der Chemie, vol. 679, pp. 34-41, 1964. https://doi.org/10.1002/jlac.19646790106
- H.S. Rzepa, "C 4 H 9 F 3 Li 1 N 1 O 3 S 1", 2015. https://doi.org/10.14469/ch/191545
- H.S. Rzepa, "C 5 H 11 F 3 Li 1 N 1 O 3 S 1", 2015. https://doi.org/10.14469/ch/191553
- H.S. Rzepa, and H.S. Rzepa, "C 5 H 17 F 3 Li 1 N 1 Ni 1 O 3 P 2 S 1", 2015. https://doi.org/10.14469/ch/191554
- H.S. Rzepa, "C 5 H 17 F 3 Li 1 N 1 Ni 1 O 3 P 2 S 1", 2015. https://doi.org/10.14469/ch/191536
- H.S. Rzepa, "C5H17F3LiNNiO3P2S", 2015. https://doi.org/10.14469/ch/191550
- H.S. Rzepa, and H.S. Rzepa, "C 5 H 17 F 3 Li 1 N 1 Ni 1 O 3 P 2 S 1", 2015. https://doi.org/10.14469/ch/191555
- H.S. Rzepa, "C 3 H 12 Ni 1 P 2", 2015. https://doi.org/10.14469/ch/191547
- H.S. Rzepa, "C3H12NiP2", 2015. https://doi.org/10.14469/ch/191546
- H.S. Rzepa, "C 3 H 12 Ni 1 P 2", 2015. https://doi.org/10.14469/ch/191541
- H.S. Rzepa, "C 3 H 12 Ni 1 P 2", 2015. https://doi.org/10.14469/ch/191540
- H.S. Rzepa, "C3H12NiP2", 2015. https://doi.org/10.14469/ch/191548
- H.S. Rzepa, "C 3 H 12 Ni 1 P 2", 2015. https://doi.org/10.14469/ch/191542
- H.S. Rzepa, "C 3 H 12 Ni 1 P 2", 2015. https://doi.org/10.14469/ch/191537
- H.S. Rzepa, "C3H12NiP2", 2015. https://doi.org/10.14469/ch/191538
- Buchalski, P.., Grabowska, I.., Kaminska, E.., and Suwinska, K.., "CCDC 650794: Experimental Crystal Structure Determination", 2008. https://doi.org/10.5517/ccpv6c2
- P. Buchalski, I. Grabowska, E. Kamińska, and K. Suwińska, "Synthesis and Structures of 9-Nickelafluorenyllithium Complexes", Organometallics, vol. 27, pp. 2346-2349, 2008. https://doi.org/10.1021/om701275u
Tags: activation free energy barriers, Cambridge, energy, energy relative, nickel-carbene product