No doubt answers to the question posed in the previous post are already being obtained by experiment. Just in case that does not emerge in the next day or so, I offer a prediction here.
The methodology is the same as before, and I have not tried to look for new isomeric forms compared with the structures found with HCl. The method as before is DFT-based: ωB97XD/6-311++G(2d,2p). In the table below, I am recording the halogen-H distance and the distance from the same H to oxygen. You might also observe a more general principle here; first calibrate the method you intend to use with a system where there is an experimental answer. If the two match, use the same method to predict (extrapolate) to systems as yet unmeasured.
| F | Cl | Br | ||||
|---|---|---|---|---|---|---|
| n | F-H, Å | H-O | Cl-H | H-O | Br-H | H-O |
| 1 | 0.937 | 1.702[1] | 1.300 | 1.857 | 1.438 | 1.912[2] |
| 2 | 0.951 | 1.631[3] | 1.322 | 1.728 | 1.463 | 1.754[4] |
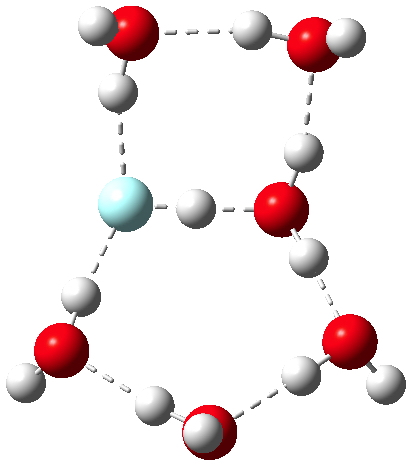
| 3 | 0.967 | 1.532[5] | 1.351 | 1.579 | 1.506 | 1.554[6] |
| 4 | 0.972 | 1.504[7] | 1.387 | 1.470 | 2.032 | 1.028[8] |
| 5 | 1.043 | 1.329[9] | 1.841 | 1.034 | 2.039 | 1.021[10] |
| 6 | 1.067 | 1.283[11] | 1.880 | 1.023 | 2.073 | 1.013[12] |
From the bond distances, one notices that “ionisation” is an abrupt discontinuous event, happening for four molecules with HBr, five molecules with HCl and more than six molecules with HF. This nicely parallels the pka values: HBr (pKa = -9.0) < HCl (pKa = -6.0) << HF (pKa = +3.1).
It is good to see that such a process modelled on the nanoscale using just a few discrete molecules can map onto the macroscopic scale of solutions.
Postscript: If you check on the structures of these systems (click on the pictures in the previous post) you will see that the discontinuous ionisation event occurs in a bicyclic system, with the water forming two separate rings. Evidence that this really is the structure of microsolvated species has recently been put forward[13].
Acknowledgments
This post has been cross-posted in PDF format at https://orcid.org/0000-0002-8635-8390
References
- Henry S Rzepa., "H 3 F 1 O 1", 2015. http://dx.doi.org/10.14469/ch/190911
- Henry S Rzepa., "H 3 Br 1 O 1", 2015. http://dx.doi.org/10.14469/ch/190907
- Henry S Rzepa., "H 5 F 1 O 2", 2015. http://dx.doi.org/10.14469/ch/190910
- Henry S Rzepa., "H 5 Br 1 O 2", 2015. http://dx.doi.org/10.14469/ch/190909
- Henry S Rzepa., "H 7 F 1 O 3", 2015. http://dx.doi.org/10.14469/ch/190912
- Henry S Rzepa., "H 7 Br 1 O 3", 2015. http://dx.doi.org/10.14469/ch/190913
- Henry S Rzepa., "H 9 F 1 O 4", 2015. http://dx.doi.org/10.14469/ch/190915
- Henry S Rzepa., "H 9 Br 1 O 4", 2015. http://dx.doi.org/10.14469/ch/190916
- Henry S Rzepa., "H 11 F 1 O 5", 2015. http://dx.doi.org/10.14469/ch/190918
- Henry S Rzepa., "H 11 Br 1 O 5", 2015. http://dx.doi.org/10.14469/ch/190919
- Henry S Rzepa., "H 13 F 1 O 6", 2015. http://dx.doi.org/10.14469/ch/190928
- Henry S Rzepa., "H 13 Br 1 O 6", 2015. http://dx.doi.org/10.14469/ch/190917
- C. Pérez, J.L. Neill, M.T. Muckle, D.P. Zaleski, I. Peña, J.C. Lopez, J.L. Alonso, and B.H. Pate, "Water–Water and Water–Solute Interactions in Microsolvated Organic Complexes", Angewandte Chemie International Edition, vol. 54, pp. 979-982, 2014. http://dx.doi.org/10.1002/anie.201409057
Just an observation: The pKa of HF indicates that the lowest (free) energy state in the bulk is undissociated HF, so no amount of H2O molecules should cause dissociation of HF based on the assumptions you make (i.e. that you have found the lowest E solution and that E approximates G)
Yes, there are assumptions in moving from nanoscale to macroscale. One has to be careful with standard states (which for these calculations would emerge by default as 1 atmosphere pressure, or 0.044M). And I suppose that ΔE does not approximate ΔG, since entropy must also play a very major role here. Also, since the original experiment that reported the ionisation of HCl by water molecules was conducted in the gas phase, I have not included any sort of further continuum model here; these are gas phase calculations. So it is a slight stretch to compare them with solution properties such as pKa!
I have not quoted any energies deliberately for these reasons; calculating a pKa for a solution is non-trivial.
I agree. My main point is that the lowest energy structure of HF(H2O)n will be non-dissociated HF for any value of n because the pKa of HF is positive,
Entropy will favor dissociation and should therefore not change this conclusion.
It is always worth trying to unpick the semantics of any situation. In this case, what do we mean by non-dissociated HF? The simple response might be covalent HF rather than ionic H3O(+).F(-), and pKa assumes you can measure concentrations of both such species.
Now, for both HF and its ionic form to be present (and here again one has to define for how long for), one must presume what is called a double-minimum potential well, at the bottom of one well there is sitting HF and in the other the ionic form, separated by some sort of barrier that defines the lifetime of each species. The relative depth of each well (+ ZPE) defines the equilibrium constant, and hence pKa.
But suppose that HF + H2O eventually settles into a single well, where the HF is neither fully non-ionic or fully ionic, but something in between. Any measure that attempts to define say the concentration of ionic species (a conductivity) may give us a strange answer to the single well. That species might lead to “slight” conductivity, which could lead us to believe the species is non-dissociated.
I am not saying that this is the case for HF + H2O, rather that one might conceive of models where the meaning of an apparent measurement of a pKa may not be what it seems.
Thus the pKa of water is 15.7, at which the instantaneous concentration of [OH-] or {H3O+] are both ~10^^-7 M. But the ion-pair, surrounded by covalent water molecules, is probably not a minimum in the potential energy surface. Proton transfers, via a chain possibly, have zero barriers, and the ion-pair may only have a lifetime of a vibration or so. This is probably long enough to result in measurable conductivity. I am sure this explaination is of itself very simplistic, and possibly even wrong in parts. I am just using it to say that what goes on in such solutions may not correspond to the simple pictures taught in elementary chemistry.
Well, I stand corrected! Turns out you are absolutely right: http://en.wikipedia.org/wiki/Hydrogen_fluoride#Acidity
I learned something today!
I should perhaps correct what I wrote in the post “that “ionisation” is an abrupt discontinuous event, happening … for more than six molecules with HF. ”
Whilst it is clearly discontinuous for HCl, HBr and HI, it might not be discontinuous for HF. instead, the system might settle into a half-way house of a single-well minimum. I wonder if the apparatus used to detect the HCl+H2O clusters would be happy with HF inside it?
I would also add that the original experiment which catalysed this series of posts, that of determining a sudden increase in the dipole moment of small HCl+H2O clusters is also open to some ambiguity. I managed to find a cluster of HCl+5H2O which had a small dipole moment, despite being largely dissociated. So it would not be detected.
Here is a crystal structure search of some HF compounds, showing the H-F distance along the y-axis and the distance from the H to any group N,O,F or Cl along the x-axis.
It shows a natural asymmetry in the compounds, with the H-F distance short, and the H…X distance longer, very much as seen in the table above.
Here is the same search repeated, but this time only for F…H…F compounds. The maximum (red) is symmetric, but there is a long asymmetric tail.
Following on from the FHF plot above, here is a OHO plot, but with the specifics of H2OHOH2 (in other words, the O2H5(+) cation).
Notice again the prominent diagonal line, with the hotspot at the symmetric H, but a long asymmetric tail again. I do have to point out that crystallographic determinations of H positions using X-rays would tend to locate the H at the centre, even if the actual potential were asymmetric, since the barrier for interconverting them is small.
And I end this comment with the crystal structure of O4H9+, which also shows asymmetric OHO positions.