Quite a few threads have developed in this series of posts, and following each leads in rather different directions. In this previous post the comment was made that coordinating a carbon dication to the face of a cyclopentadienyl anion resulted in a monocation which had a remarkably high proton affinity. So it is a simple progression to ask whether these systems may in turn harbour a large affinity for binding not so much a H+ as the next homologue He2+?

Inventing the Helium bond
This possibility is explored with the series X=Be, B, C (tetramethyl substituted, resulting in neutral, +1 and +2 systems overall). The first two emerge as stable in terms of having all positive force constants for C4v symmetry; the last emerges as a transition state and is not discussed further. The specific system X=B has a B-He bond length of 1.317Å/B3LYP/6-311G(d,p), 1.305Å/B3LYP/Def2-QZVPP and 1.290Å/double-hybrid RI-B2GP-B2PLYP/TZVPP, which does seem as if it might be typical of a single bond between these two elements. The ρ(r)B-He AIM value (B3LYP/6-311G(d,p) is 0.069 au, and νB-He of 713 cm-1 (727 for Def2-QZVPP basis) makes it about one third the strength of a C-H bond. The disynaptic basin for the B-He region integrates to 1.99 electrons, whilst the four B-C basins correspond to 1.22 electrons each.
| X | Charge | ρ(r) X-He | C-B ELF integration |
νX-He, cm-1 | Repository |
| Be | 0 | 0.031 | 1.10 | 484 | 10042/to-2443 |
| B | 1 | 0.069 | 1.22 | 713 | 10042/to-2444 |
| C | 2 | 0.026 | – | 136 | 10042/to-2445 |
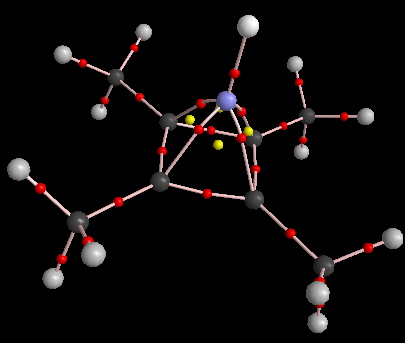 AIM for X=B-He. Click for 3D |
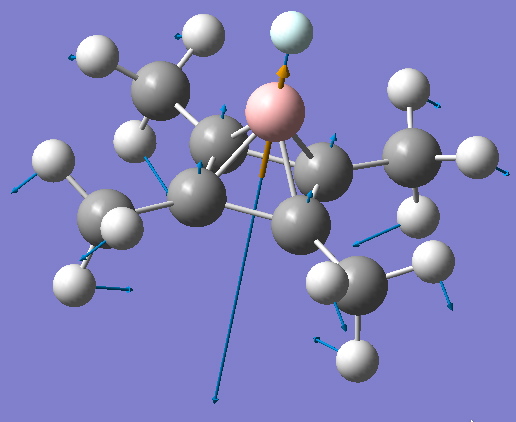 B-He stretching mode. Click to vibrate |
We can conclude that for X=B, this species exhibits not only a pentavalent boron atom, but a monovalent helium atom. The latter bond may indeed be amongst the strongest ever proposed for this element in a ground state, and indeed perhaps is even viable as a solid crystalline compound rather than merely existing in the gas phase. The Cambridge crystal database contains no entries for He or Ne, not even as an encapsulated clathrate (although crystal structures of such complexes for Kr and Ar are known). Theoretical studies of the rare gases in endohedral fullerene-like cages (DOI: 10.1002/chem.200801399) predict that under these compressed circumstances e.g. two helium atoms can approach each other to 1.265Å or less (see also DOI: 10.1002/chem.200700467) but these close approaches were not considered to be chemical bonds as we think of them. Perhaps Merino, Frenking, Krapp and co’s search for the chemistry of helium (they had found it earlier in the gas phase excited states of their molecules, DOI: 10.1021/ja00254a005) might be realised for the ground state of the system described here.
Tags: AIM, bonding, Cambridge, chemical bonds, double-hybrid, ELF, gas phase, Hypervalency, hypervalent, Interesting chemistry, Krapp and co
[…] Henry Rzepa Chemistry with a twist « Uncompressed Monovalent Helium […]