I was intrigued by one aspect of the calculated transition state for di-imide reduction of an alkene; the calculated NMR shieldings indicated an diatropic ring current at the centre of the ring, but very deshielded shifts for the hydrogen atoms being transferred. This indicated, like most thermal pericyclic reactions, an aromatic transition state. Well, one game one can play with this sort of reaction is to add a double bond. This adds quite a twist to this classical reaction!
The original di-imide reduction can be viewed as a six-electron process; one that fits the 4n+2 aromaticity rule. In fact, this is a specific instance of a more general topological rule, first proposed in 2008, which suggests that for 4n+2 electron thermal reactions, the electronic topology conforms to that of a Möbius link, for which the so-called linking number Lk is even (o, 2, 4, etc). For systems in which 4n electrons participate, such as the homologated example above, the rule changes to the topology of a Möbius knot, for which the linking number is odd (1, 3, etc)[1]. One interesting consequence of all this topology is that all the systems for which Lk > 0 are chiral (achiral benzene is thus seen as an exception rather than the norm of aromaticity)[2].
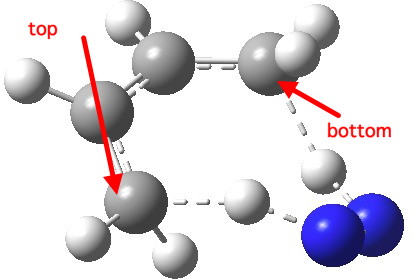
Transition state for 8-electron di-imide reduction. Click for 3D.
The calculated transition state for this reaction is shown above. As befits a torus knot, the two hydrogen atoms are transferred to opposite faces of the butadiene; an antarafacial mode. Now, to be fair the alternative mode in which the hydrogens are delivered suprafacially to just a single alkene is 26.1 kcal/mol lower in free energy. We might conclude that di-imide does not reduce butadiene in this manner, and that getting an experimental example of such stereochemistry might be a challenge!
What of the aromaticity of this Möbius version? The NICS(0) at the ring critical point is -15.7 ppm, whilst the shieldings of the transferring hydrogens are +14.0 ppm. So just like its 4n+2 electron counterpart, this Möbius di-hydrogen transfer reaction also proceeds through an aromatic transition state.
References
- S.M. Rappaport, and H.S. Rzepa, "Intrinsically Chiral Aromaticity. Rules Incorporating Linking Number, Twist, and Writhe for Higher-Twist Möbius Annulenes", Journal of the American Chemical Society, vol. 130, pp. 7613-7619, 2008. https://doi.org/10.1021/ja710438j
- C.S. Wannere, H.S. Rzepa, B.C. Rinderspacher, A. Paul, C.S.M. Allan, H.F. Schaefer, and P.V.R. Schleyer, "The Geometry and Electronic Topology of Higher-Order Charged Möbius Annulenes", The Journal of Physical Chemistry A, vol. 113, pp. 11619-11629, 2009. https://doi.org/10.1021/jp902176a
Tags: free energy, Möbius knot, Möbius link, pericyclic, Reaction Mechanism