Here is a challenge: what is the longest C-C bond actually determined (in which both carbon termini are sp3 hybridised)? I pose this question since Steve Bachrach has posted on how to stabilize long bonds by attractive dispersive interactions, and more recently commenting on what the longest straight chain alkane might be before dispersive interaction start to fold it (the answer appears to be C17).
A search of the Cambridge database (the following conditions apply; structure determined at less than 160K, no errors, no disorder, and a minimum separation of 1.7Å) reveals only 7 entries longer than 1.7A. The reference codes for these are FIBBOI, HOLKOI, LAGHOQ01, RIRTUH, RUNHOY, XIQRIZ, BATSIA, the latter having a value of 1.731Å (T=123K, R=4.44%, DOI: 10.1246/cl.2012.541). Of course, a very close inspection of the crystallography would also be needed to determine if these values are to be taken at face value.
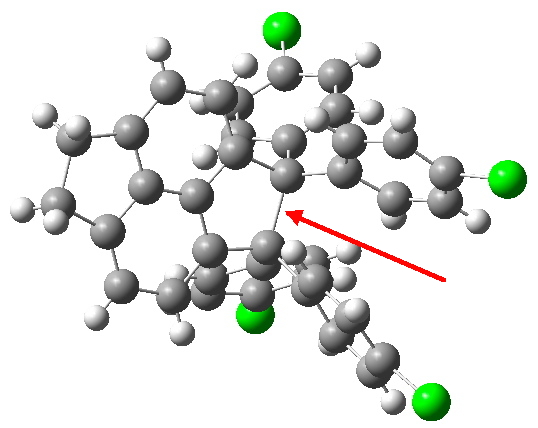
BATSIA. Click for 3D model.
If one relaxes the temperature constraint for measurement, CAZFUE01 has a claimed C-C length of 1.99Å (R=4.5%, DOI: 10.1246/cl.2012.541). This however represents the so-called frozen transition state for a [3,3] sigmatropic rearrangement. A ωB97XD/6-311G(d,p)/SCRF=dichloromethane calculation seems to indicate that this species is in fact a transition state for the [3,3] reaction, and hence that the crystal structure may in fact correspond to the mean position of the two end points of the reaction, and so be an artefact. More on this in a later post.
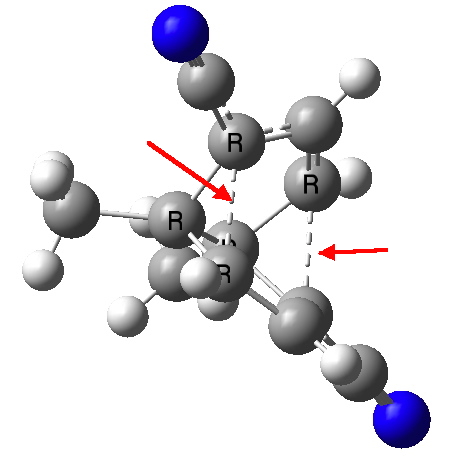
CAZFUE. Click for 3D model.
And I suppose one should ask what the shortest such single bond is as well as the longest. This is currently claimed to be 1.436Å for KAVKUO (10.1002/anie.200501605) and 1.434Å for WUDKAH (10.1016/S0040-4020(02)00695-6). The origins of this contraction also deserve to be more fully explored in a later post.
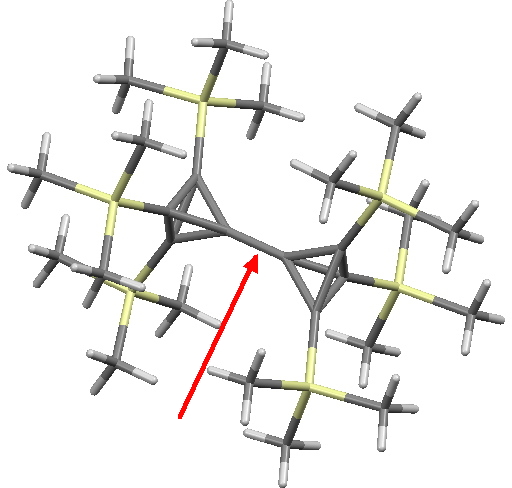
KAVKUO. Click for 3D
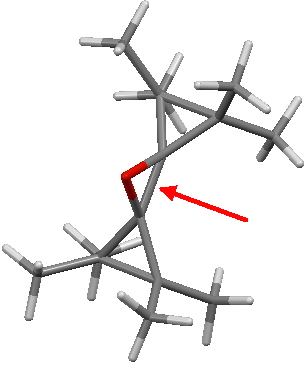
WUDKAH. Click for 3D.
Certainly, the (sp3)C-C(sp3) bond does seem to be capable of a large range of values ranging from 1.44 to at least 1.73 and possibly 1.99Å.
Tags: Cambridge, Steve Bachrach
Henry, I don’t think your title asks the question meaningfully. What is a bond?
The 1,3-CC separation is 2,5Å between the ca. sp3 methyl carbons in e.g., propane. Is this a bond? The C’s in the “CC bonds” you illustrate surely are not “sp3 hybridized (especially in KAVKUO and WUDKAH, where they are ca. “sp”). The CC bond length is about 2Å in the ethane radical cation, and I’ve computed similar “one electron” C-C bond lengths up to 2.5 Å even in neutral molecules (containing B’s instead of C+’s). These don’t have “sp3” hybridized C’s, but neither do the C’s comprising any “stretched” C-C bond, since they must have partial singlet diradical character. And why restrict the search to X-ray data, interesting as it may be? Thats not specified by your title question.
I will have a post soon on another paper by Schreiner where he reports the dimer of two radicals formed from an extended diamond-like structure that has the “bond length” over 4 Å – yet the two halves are held together by over 20 or so kcal/mol
By single bond, I meant a two-electron Lewis bond between two four-coordinate carbon atoms. The term sp3 in this sense was indeed too loose. Four coordinate is better (I agree that carbon in three-membered rings is often described as sp5+sp hybridised!). It is also interesting to speculate at what elongated distance a C-C two electron bond stops being such, and becomes a loosely interacting biradical. I would think not yet at 1.73Å, but quite possibly at say 2Å. I would also note that the Cambridge database is formally searched only for integer bond orders (this is how one specifies a search), and even that depends on how the deposited data is described by the curators. I have noted previously that some types of compound containing eg S…S interactions are sometimes recovered from the database by a search specifying a S-S single bond, and sometimes by specifying no S-S bond (to find all the entries, one has to do a boolean combination of two separate searches, with an OR). If one did not apply such a search mask, searches would take many times longer (i.e. based purely on interactomic distances). So this really is a practical expedient, rather than one based on a deeper definition of what a bond really is!
Compound WUDKAH was deliberately ambiguous. Written as a reactant for a [3,3] sigmatropic shift, the terminal carbons are sp2, becoming sp3 only in the product. So at the half-way stage, they must be intermediate (or indeed biradical, and hence forming two 1-electron bonds instead of one 2-electron bond). The same ambiguity applies to e.g. two-electron-3-centre carbocations such as the norbornyl cation. There one has C-C distances of around 2Å, and again hybridisation that does not fall nicely into the classical sp2, sp3 categories.
One does tend to assume that bonds have to be quantised into single, double triple, with perhaps half-values corresponding to 0.5, 1.5, 2.5. But the Cambridge data base (lets face it, there are more than 600,000 molecules and many more bonds described there by experimental data, a far wider spread than other spectroscopic techniques) does teach us that even the apparently straightforward two-electron single bond may be a fuzzier concept than Lewis would have imagined, spanning lengths ~1.43-1.73Å.
Replying to Steve, I recollect seeing a talk once about Na2, in which the two sodium atoms were said to interact at a distance of several hundred Å, and were described as a Rydberg bond.
So the range of bonds defined by a bond dissociation energy of ~20 kcal/mol can range from F-F (~1.45 Å) to 4Å! I guess the answer is that these two extremes are described by very different quantum mechanics. F-F is a classical example of a charge-shift bond, where the covalent component is actually repulsive, and of course the 4 Å one is presumably a strongly interacting biradical (but perhaps not through space but through other bonds?).
We might have thought we understood even the classical two-centre two-electron Lewis bond, but apparently not!
With the caveats above noted by Paul, I have found another structure (MAXKIG and possibly MAXKUS (DOI: 10.1021/cg050064r) that appears to extend the upper range of the length of a C-C single bond to 1.77-1.79Å (this observation was actually noted in DOI: 10.1021/ed800058c). These are a set of hexaphenylethane derivatives, and the authors note how very conformationally dependent the precise length is.
[…] Henry Rzepa Chemistry with a twist « What is the range of values for a (sp3)C-C(sp3) single bond length? […]
Oliva, Annan, Schleyer, Vinas, and Teixidor, JACS, 2005, 127, 13538 report “tunable” carborane systems whereby C-C distances can be varied from 1.626 to 2,638Å, simply by changing substituents. “There is no abrupt discontinuity over the entire range of CC lengths.” In short,one can get any CC distance one wishes in this ~1 Å range! Deciding when a “CC bond ” stops being a “CC bond” along this range is a matter of definition and semantics. Obviously, a “CC single bond” has an increasingly smaller “bond order” upon being stretched continuously. There is no “breaking point” definable by distance or energy. I have computed such “CC bond-breaking” (energy vs. CC distance) plots for ethane dissociation into two CH3 radicals and the for the similar disruption of hexamethyl ethane into two t-butyl radicals and was surprised by the still appreciable “CC bond enegy” remaining at large (e.g., 2.5 Å) CC separations.
Thanks for point out this fascinating study Paul. I had been aware that one can tune an S-S bond, but not that the effect also applied to e.g. C-C. The examples you cite are carboranes, or electron deficient systems, and so I guess when there is a deficit of electrons, there must be several alternatives (indeed a continuum) for where they may end up.
Indeed, upon reflection, it may be quite a general phenomenon. I recollect noting that a C-S bond can be tuned across about two bond orders depending on the attached substituents (and this is not even an electron deficient system).