Organic chemists have been making (more or less pure) molecules for the best part of 180 years. Occasionally, these ancient samples are unearthed in cupboards, and then the hunt for their origin starts. I have previously described tracking down the structure of a 120 year-old sample of a naphthalene derivative. But I visited a colleague's office today, and recollected having seen a well-made wooden display cabinet there on a previous visit. Today I took a photo of one of the samples:
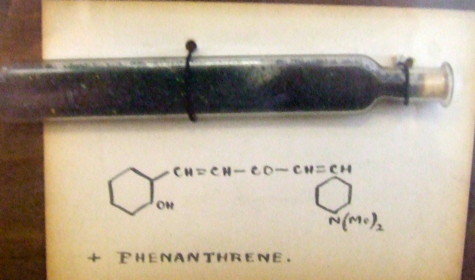
One of the "Hofmann" collection.
No date, no name, but a structure! As I noted before, when it comes to structures, you have to research the conventions (and numbering) used at the time. Thus note the apparent cyclohexane rings, the N(Me)2 group and the lack of stereochemistry around the alkenes. The former dates the sample to before 1950, whilst the use of Me to mean methyl puts it in the 20th century. Which is shame, since it had been known as the "Hofmann" collection, meaning some sort association with August von Hofmann, the first professor of organic chemistry in the UK, who occupied that position from 1845-1864. Samples that old are very rare. The one above by the way is very deep green (the photo does not do it justice), and very crystalline! Tracing the history of where the display cabinet might have been did indeed reveal that it probably started its life at the same institute as Hofmann was working in (and where I now work), but little more than this was known about it.
A search of the Beilstein database (nowadays known as Reaxys) revealed a collection of samples corresponding to the above structure (with benzenes of course, not cyclohexanes), but co-crystallised with different molecules, and dating from 1921. These were known as the Heilbron collection, and this was encouraging, since Heilbron was indeed a successor to Hofmann, being active in the 1920s. During his career, he and his students probably made 100s, if not 1000s of compounds, so why did they go to the considerable expense of having beautiful wooden cases built to house these particular samples? Probably because the basic colour varied from yellow to black (perhaps 400nm difference in λmax) and for which they had no explanation! So, much like some people are cryofrozen in the hope an advanced civilisation might bring them back to life in the future, these samples were mounted in a display cabinet in the hope that someone would find out the origins of their variable colour.
Well, in 1984 (some 63 years after the event)[1] researchers at the lsrael Institute of Technology, Haifa, came upon the 1921 article (but not the samples; if they read this they might be amazed that these still exist!), repeated (most of the) syntheses, and determined the crystal structure of three of the molecules (but conspicuously not the one above). One 3D structure is shown below. The colours were ascribed to charge-transfer interactions between the components of the molecules.
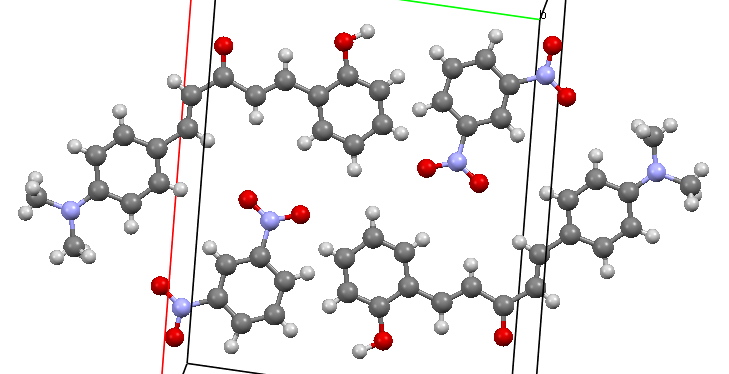
DADZIR. Click for 3D
As I noted previously, it is well worth preserving chemical samples for future generations (and sometimes that generation is 120 years in the future!). Sadly, health and safety aspects (real or imagined) mean that such collections are being lost to posterity at an every increasing rate. Soon, there may be no collections of old chemicals left. That would be indeed a loss to science. So if you know of a lovingly preserved case of old chemicals, go take a look at it. And if it's in danger of being put in the skip, then rescue it. There is no telling what may be scientifically interesting about it.
References
- F.H. Herbstein, M. Kapon, G.M. Reisner, and G.M. Rubin, "Crystallographic studies of the molecular complexes ofE,E-1[p-dimethylaminophenyl]-5-[o-hydroxyphenyl]-penta-1,4-dien-3-one 9DHDK) with chloroform (1:0.4),m-dinitrobenzene (1:1) andp-dimethylaminobenzaldehyde (1:1); the Heilbron complexes", Journal of Inclusion Phenomena, vol. 1, pp. 233-250, 1984. https://doi.org/10.1007/bf00656759
Tags: August von Hofmann, chemicals, detective, first professor, Haifa, Historical, Institute of Technology, professor of organic chemistry in the UK, Technion, United Kingdom, worth preserving chemical samples
[…] (10.1039/CT8875100258 and 10.1039/PL8900600095). Let me set the scene by reminding of an earlier post showing the structure of a bis(stilbyl)ketone, dated 1921. The two aromatic groups (yes, they […]