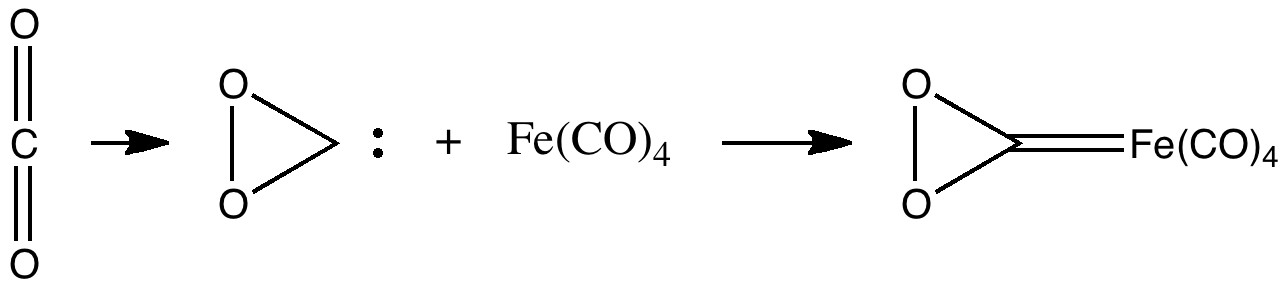The (hopefully tongue-in-cheek) title Mindless chemistry was given to an article reporting[1] an automated stochastic search procedure for locating all possible minima with a given composition using high-level quantum mechanical calculations. “Many new structures, often with nonintuitive geometries, were found”. Well, another approach is to follow unexpected hunches. One such was described in the previous post, and here I follow it to one logical conclusion.
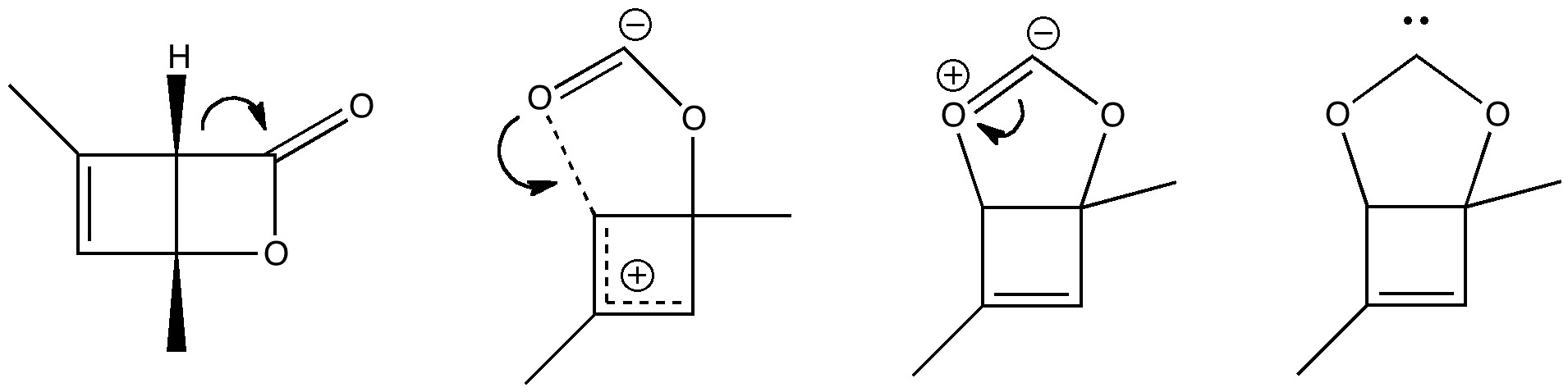
The train of thought started with the recent speculation upon a zwitterionic intermediate in the photolysis of a dimethyl-pyrone. Closure of this is likely to require a very low barrier, and this leads to a bicyclic species, which could be written as a carbene. One then asks if carbon dioxide itself could be so represented? If so, could that carbene be stabilised with a metal, as below? A reality check, as noted in the earlier post, is that a similar complex with iron tetracarbonyl is known, and appears to be stable.
Enter quantum mechanics, which will tell us exactly how stable. Firstly, the spin state of the complex has to be determined, and it turns out the singlet (low spin) is lower than either the triplet (medium spin) or quintet (high spin) states. It took around five minutes (ωB97XD/6-311G(d) ) to establish that the free energy of the reaction between carbon dioxide and iron tetra carbonyl is endothermic in free energy by ~100 kcal/mol. So no sequestration of CO2 by iron carbonyl then!
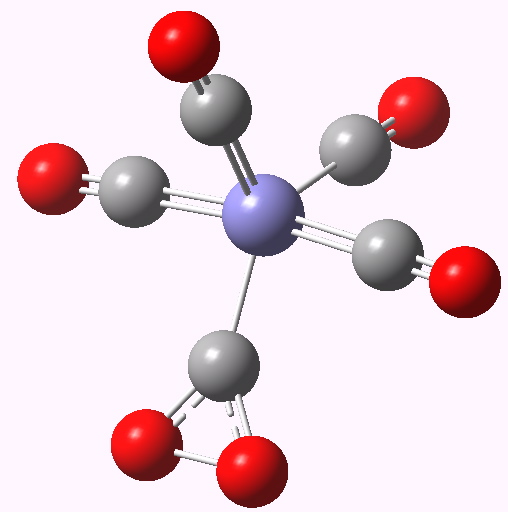
Iron tetra carbonyl-carbon dioxide complex. Click for 3D
As a scientist, I always find it fascinating how one can jump from one topic to a completely different one in just a few steps. But one always needs reality checks in doing so! Perhaps automated mindless searches (bounded by quantum mechanical reality checks) will perhaps one day come up with something really important. All us humans have to do is recognise this when it happens.
References
- P.P. Bera, K.W. Sattelmeyer, M. Saunders, H.F. Schaefer, and P.V.R. Schleyer, "Mindless Chemistry", The Journal of Physical Chemistry A, vol. 110, pp. 4287-4290, 2006. https://doi.org/10.1021/jp057107z
Tags: automated stochastic search procedure, free energy, metal, scientist, Tutorial material