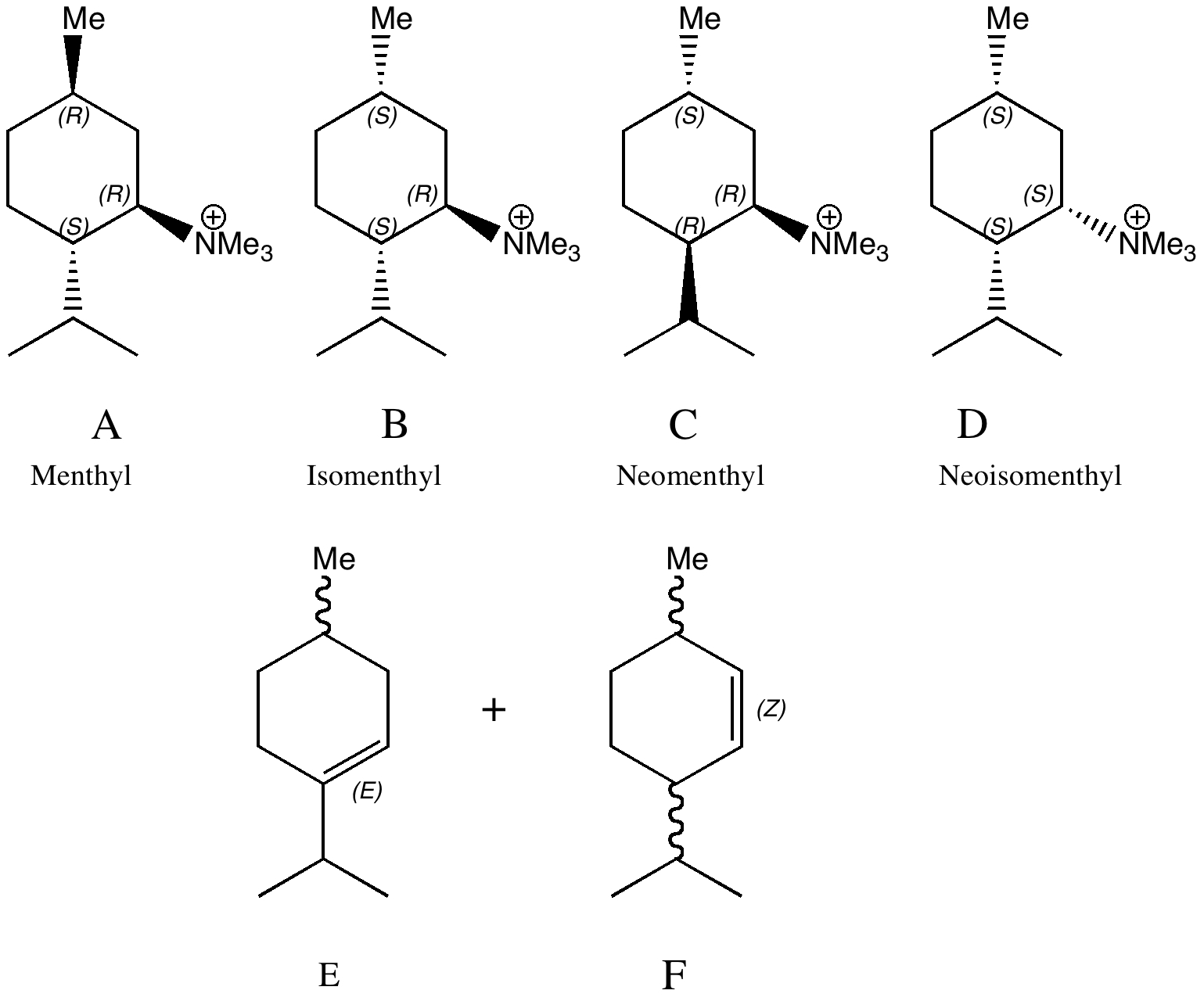
Conformational analysis comes from the classical renaissance of physical organic chemistry in the 1950s and 60s. The following problem is taken from E. D. Hughes and J. Wilby J. Chem. Soc., 1960, 4094-4101, DOI: 10.1039/JR9600004094, the essence of which is that Hofmann elimination of a neomenthyl derivative (C below) was observed as anomalously faster than its menthyl analogue. Of course, what is anomalous in one decade is a standard student problem (and one Nobel prize) five decades later.
One can pose two questions about these systems:
- What is the expected product formed by reaction of A-D; E or F or both?
- Can the four reactions be ranked in the order fastest to slowest (the hint that C is anomalously fast may or may not be given the students!)
Its a problem that simply requires a model to be built for its solution. And probably some hints. I give the students two:
- Each of the reactants can have two alternative chair conformations; let us call them A1 and A2 etc (although a really adventurous student might ask if any twist-boats are also possible). In general, only one of these two can react to eliminate the trimethylammonium group to form an alkene. The task is to determine which, and whether that also reveals whether E or F or both might be formed.
- The rate of reaction (all other things being equal, which they might not be) will be related to the concentration of the active conformation compared to the inactive one. So one has to decide which of the two conformations is likely to be lower in energy, and by how much. Here one can bring as many rules as you might find in the texts books (or lecture courses) to bear whilst you decide. If you are really keen, you can try building a model using suitable molecular modelling software.
So that I do not spoil your fun, I will not reveal (my) answers here, but in the next post. Try writing down answers to these two questions, and see if they agree with mine!
Tags: chair, conformational analysis, cyclic systems, energy, suitable molecular modelling software, Tutorial material
[…] Henry Rzepa Chemistry with a twist « Updating a worked problem in conformational analysis. Part 1: the question. […]