In the previous post I pondered the colour of Monastral blue (copper phthalocyanine). Something did not quite fit, and so I speculated that perhaps some oxidation of the pigment might give a new species. This species (Cambridge code FEGJOQ) comprises two parts of copper phthalocyanine, 1 part of the corresponding cation, and 1 part of triodide anion. Looking at the packing of this system, I spotted something I had seen some time ago in NaI2.Acetone, namely an infinitely long and absolutely straight chain of iodine atoms, a molecular wire if you like.
A different view shows how this wire runs down layers of the phthalocyanine. The iodines are 3.2Å apart, compared to the sum of their van der Waals radii of ~4.0Å.
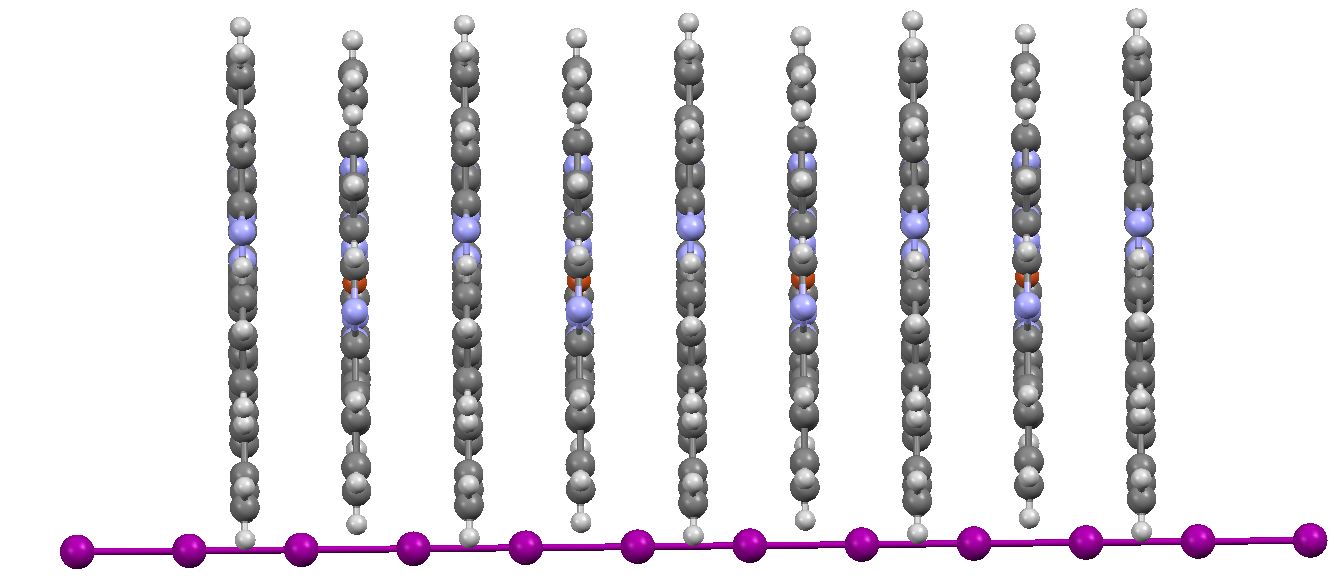
FEGJOQ, seen edge on. Click for 3D
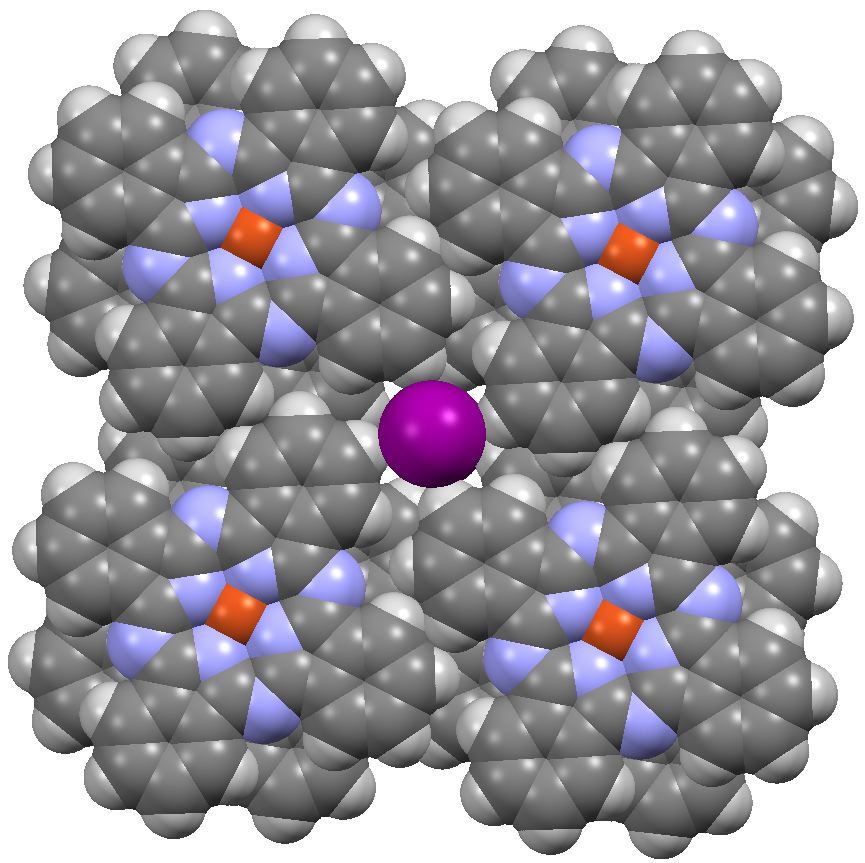
End on view, showing the filling of the channel by iodines. Click for 3D
Tags: Cambridge, molecular wire, phthalocyanine
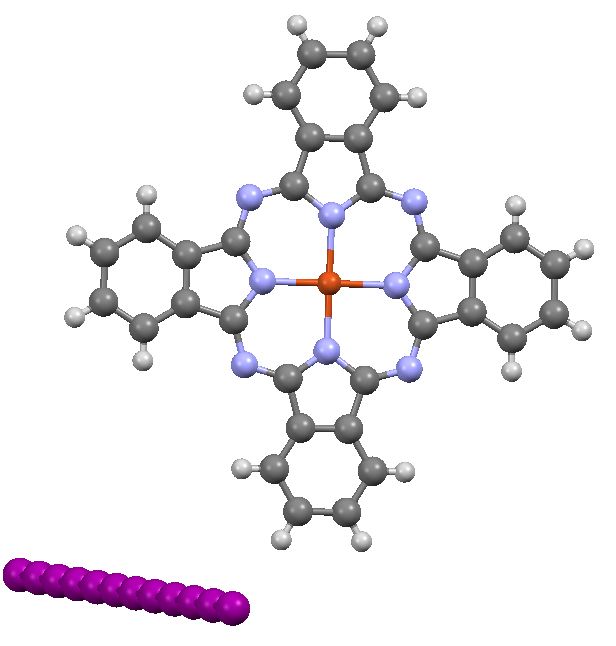
The original authors of the paper reporting this structure do not agree with your analysis of the iodines. Their 1987 JACS paper (DOI: 10.1021/ja00238a021) refers back to the isomorphous Ni species that they reported in an earlier 1980 JACS paper (DOI: 10.1021/ja00542a008). In this earlier paper they include a detailed analysis of the iodines (pages 6708 and 6709) and conclude that they have disordered chains of linked (I3)– anions. So, no continuous iodine wire, I’m afraid.
It boils down to whether linked chains of I3– are continuous or not, the key operator being linked. Another interesting question is whether the oscillation in bond length is a Peierls distortion or not.