This is a little historical essay into the electronic structure of naphthalene, presented as key dates (and also collects comments made which were appended to other posts).
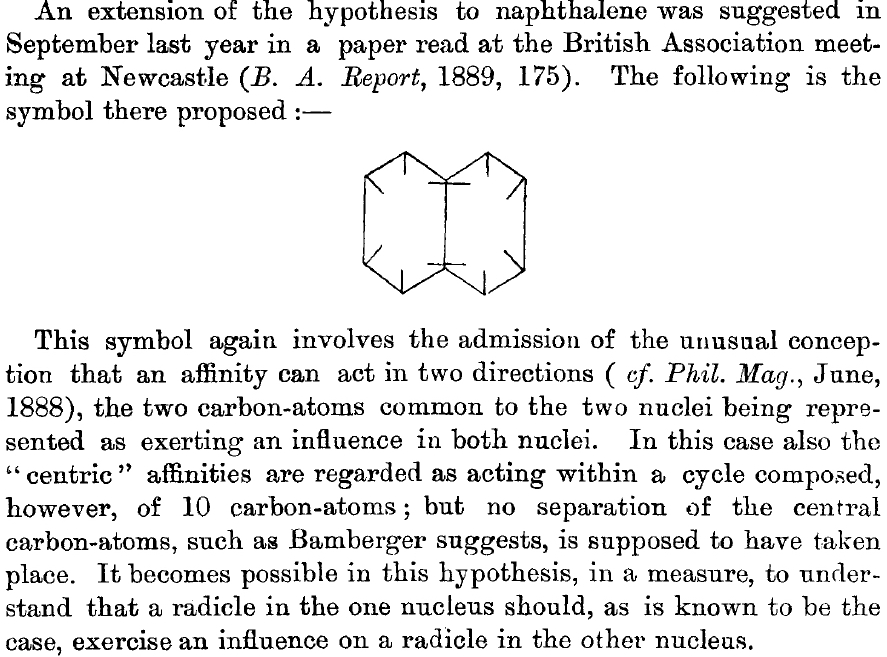
- 1890[1]: Henry Armstrong presents the following structure of naphthalene. Three words need translation into modern usage. Where he uses the word nuclei the closest translation now might be rings. Secondly, the term affinity is nowadays replaced by electron. This latter term was first coined by Stoney[2] one year after Armstrong wrote this article to mean a then hypothetical “atom of electricity”. Oddly Armstrong never updated his own usage even after Thomson actually discovered the electron in 1897. Radicle is a substituent on the ring, and the origin perhaps of the generic R used nowadays.
Notice that Armstrong talks about a cycle of ten carbons in which ten affinities/electrons act (he had previously accounted for the 22 affinities associated with what we would now call the 11 C-C σ-bonds) and is adamant that no separation of the central carbon atoms takes place as Bamburger had suggested. In modern parlance the central C-C bond has a σ-bond and he is describing a [10]annulene. The last sentence above presages the modern term delocalisation.
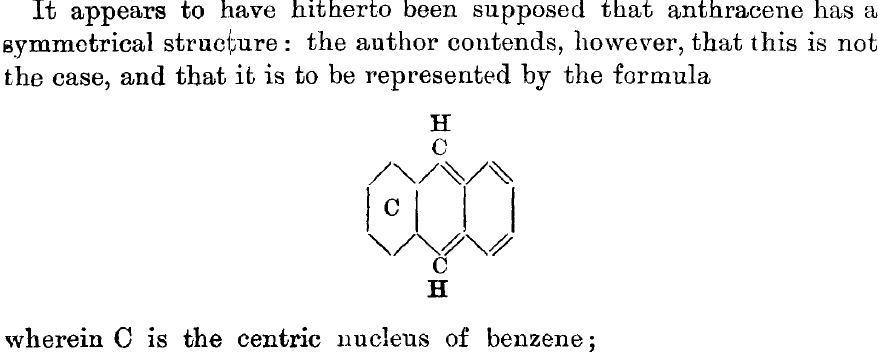
Armstrong next considers anthracene (above) and replaces the line representation of the affinities by a circle, abbreviated C (which represents six cyclic affinities, or electrons) and by four conventional double bonds, recovering 14 of what nowadays designate as π-electrons. What he does NOT do is consider the equally valid structure where his C is shown in the right hand ring, and then apply Kekule’s hypothesis to in effect average them on a chemical time scale. It is noteworthy that overall, Armstrong has discussed 6, 10 and 14 electrons, just a hint of the 4n+2 rule yet to come.
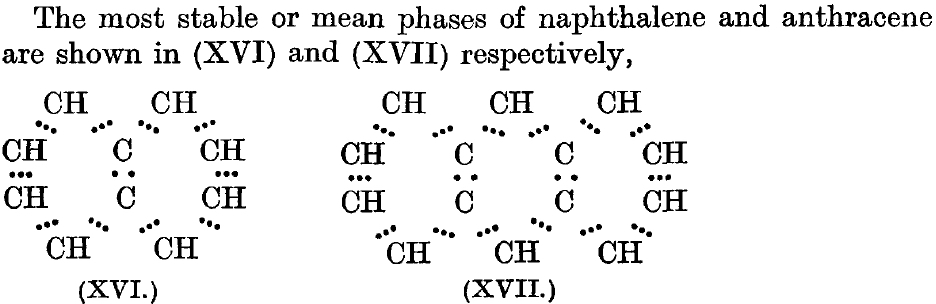
- 1922[3]:The next example comes from Robert Robinson, future Nobel prize winner, who collected all 32 electrons in naphthalene (excluding CH) into the representation show as XVL. This is an averaging (mean) that Armstrong did not do, of what we would nowadays call two resonance forms. Whereas Armstrong had clearly recognised two sets of electrons (22 and 10), this distinction is lost in this 1922 representation of the 32 ring electrons in naphthalene.
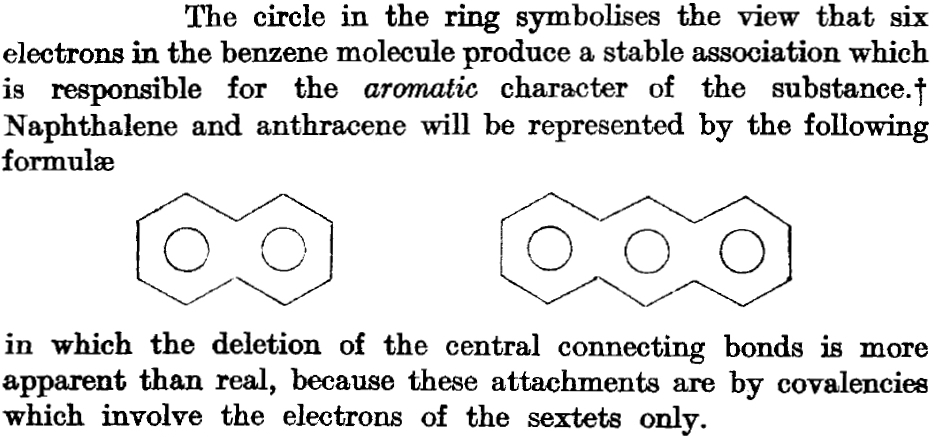
- 1925[4]Just three years later Robinson (re?)discovers the magic of six π-electrons (the term π was not yet coined) and decides to reapply it to naphthalene. Rather than average two equivalent structures, each with just six cyclic π-electrons (Armstrong’s C) he uses two such rings with twelve π-electrons. This means that he implies only 20 σ electrons (32-12=20), because to balance his count he has to remove two from the central C-C bond. When he writes that the deletion of the central connecting bond(s) is more apparent than real, he is really describing for the first time what we nowadays call a homo-π-bond, one with no underlying σ-bond (also called a suspended π-bond). On the premise one can never have too much of a good thing, he also applies this to anthracene.
- 2015: Posterity has now decided that Robinson’s 1922 effort has more or less survived and his 1925 effort has not. But one might ask whether this ill-fated suggestion could in fact inspire modern chemistry? Well, crystalline examples of such suspended π-bonds are now indeed known[5] and there are probably many more out there. I too have been inspired by the fun and games Robinson had with those two electrons;
I have forcibly removed two electrons from the system by replacing the two central carbon atoms with boron. And now playing Armstrong and Robinson’s games leaves either only an 8π periphery with a central B-B σ-bond[6] or one can raid the two B-B electrons to top the π-periphery up to 10 electrons.[7]
- The first isomer, as a 8π-electron system is according to modern knowledge antiaromatic. A ωB97XD/6-311G(d) calculation shows this is not a stable minimum, with negative energy force constants showing a twisting motion trending to a Möbius ring? It never reaches this, since further C-B bonds are ultimately formed to create an unrelated structure[8].
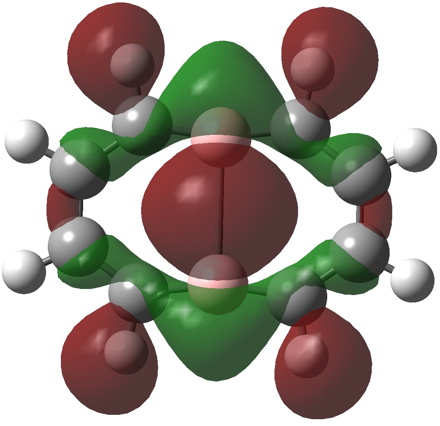
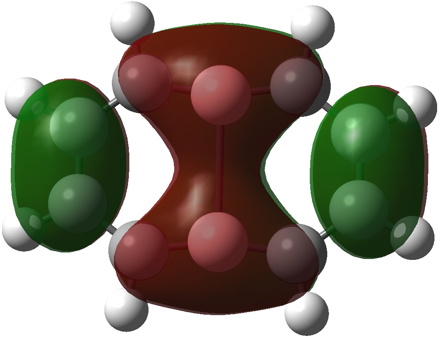
- a 10π form is 35.7 kcal/mol lower than the first and reveals five π MOs, the highest energy of which is shown below with a suspended π-bond between the two central boron atoms and a LUMO corresponding to an empty B-B σ-bond.
I hope this illustrates how science often iterates to final solutions, but that even the incorrect oscillations can still teach us chemistry.
References
- "Proceedings of the Chemical Society, Vol. 6, No. 85", Proceedings of the Chemical Society (London), vol. 6, pp. 95, 1890. https://doi.org/10.1039/pl8900600095
- G.J. Stoney, "XLIX. <i>Of the “electron,” or atom of electricity</i>", The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, vol. 38, pp. 418-420, 1894. https://doi.org/10.1080/14786449408620653
- W.O. Kermack, and R. Robinson, "LI.—An explanation of the property of induced polarity of atoms and an interpretation of the theory of partial valencies on an electronic basis", J. Chem. Soc., Trans., vol. 121, pp. 427-440, 1922. https://doi.org/10.1039/ct9222100427
- J.W. Armit, and R. Robinson, "CCXI.—Polynuclear heterocyclic aromatic types. Part II. Some anhydronium bases", J. Chem. Soc., Trans., vol. 127, pp. 1604-1618, 1925. https://doi.org/10.1039/ct9252701604
- A. Doddi, C. Gemel, M. Winter, R.A. Fischer, C. Goedecke, H.S. Rzepa, and G. Frenking, "Low‐Valent Ge<sub>2</sub> and Ge<sub>4</sub> Species Trapped by N‐Heterocyclic Gallylene", Angewandte Chemie International Edition, vol. 52, pp. 450-454, 2012. https://doi.org/10.1002/anie.201204440
- H.S. Rzepa, "C 8 H 8 B 2", 2015. https://doi.org/10.14469/ch/191378
- H.S. Rzepa, "C 8 H 8 B 2", 2015. https://doi.org/10.14469/ch/191380
- H.S. Rzepa, "C 8 H 8 B 2", 2015. https://doi.org/10.14469/ch/191379
Tags: Naphthalene, Oscillation
Interesting story! By the way, I think the meaning of “affinity” is “valency” not “electron” and this explains chemists general indifference to the discovery of electron by Thomson. Only after the Lewis theory that makes a clear link between electrons and valency the mood was changed. For Armstrong carbon atom has four affinities (valencies) and since one of the affinities is not “saturated” by its neighbor atoms in the Benzene nucleus its form the centeric arrangement for saturation. He seemingly used the same idea for naphthalene but to preserve the original idea the two junction carbon atoms now must somehow share their “single” non-saturated affinity in the two nucleus simultaneously. Ingenious idea! Regarding the ideas of Robinson in 1922, he is now thinking within framework of Lewis theory so electrons are entering into the scene. However, it is very interesting to see whether Ingold also had made any comments on Naphthalene structure since he developed his mesomerim theory before the advent of quantum mechanics and the introduction of the concept of resonance by Pauling (Natural Products 4, 61-66 (1987), Natural Products 4, 53-60 (1987), Nature 141, 314-318 (1938)). I guess with the concept of mesomerim it wouldn’t be hard to propose the correct electronic structure of naphthalene within context of the Lewis theory. The ideas of sigma and pi electrons came to scene later when Huckel started applying quantum mechanics to organic molecules (seemingly inspired by his organic chemist brother) and this has also its interesting history in relation to Robinson ideas (Nature 129, 278 (1932); 129, 937-938 (1932); 130, 273 (1932)).
We could debate whether affinity means valency or electron; in 1890 there is little to distinguish the two. Except that Armstrong does write the following:
The introduction of a radicle doubtless involves an altered distribution of the “affinity”, much as the distribution of the electric charge in a body is altered by bringing it near to another body”. Here he directly introduces electric charges, and the idea that charges influence each other. I think this brings his affinity into closer correspondence with what we now regard as the charged electron, than it does with the idea of a valency which has less clear associations with charges.
He also refers to describing his affinities with vectors (he uses the term resultants). Of course any correspondence with the modern term eigenvector to describe the distribution of “affinities” is perhaps an interpretation too far!
PS Do you happen to have any DOIs for any of the articles you cite in your comment? I could easily include them here for convenience.
Re: Ingold’s contributions. Elsewhere I have written about a young student Hilda Usherwood studying for a PhD at Imperial College, and who was also using Lewis’ ideas around 1921-1924 to formalise her ideas about the “tautomerism” of acetylene. Some time during this period she met Christopher Ingold, and married him! So it is tempting to think that Ingold might have been introduced to these ideas by Usherwood.
It is also fair to suggest that Thomson himself had ideas about benzene around 1918 or earlier, which are exactly as Robinson shows in 1922. A lot of research conducted during the great war remained unpublished during that period, and only emerged after the war, sometimes years after the discovery.
Finally, is it Hückel or Mulliken who coined the descriptors σ, π, δ and φ? I think Mulliken formalised the latter two, but was it Hückel to whom we own the former labels?
These are the “doi”s:
Natural Products 4, 61-66 (1987) DOI: 10.1039/NP9870400061
Natural Products 4, 53-60 (1987) DOI: 10.1039/NP9870400053
Nature 141, 314-318 (1938) doi 10.1038/141314a0
Nature 130, 273 (1932) doi:10.1038/130273a0
Nature 129, 937-938 (1932) doi:10.1038/129937a0
Nature 129, 278 (1932) doi:10.1038/129278a0
There is no question that in late 19 century people was aware of the electric nature of matter and there are (yet tentative) theories like that of Arrhenius that attributes electric charges to atoms and molecules but there is a long way between this vague notion of “electrified matter” and a clear understanding that universally, sole negative charge carriers in atoms and molecules are electrons and the charge is quantized. Also, we must not forget that the idea of nuclear atom and concentration of the positive charge in the nucleus was appeared a decade latter through the works of Rutherford. It would be quite reasonable to assume before nuclear atom model that positive charge may also have an active role in chemistry (for instance to have their own arrangements like electrons). So, in my opinion it is an “over-interpretation” to equate affinity with electrons in the late of nineteenth century.
I was not aware of Ingold’s love affairs and their role in his science! Interesting! Finding such brides seems to be hard now, at least in Iran!!!
I was also not ware of Thomson’s work though I guess he was not in main stream physics after 1910…
Indeed, I guess it was Mulliken. I just meant it was Huckel how studied the pi electron systems of organic molecules in detail. I guess he didn’t used sigma and pi terminology. I am not sure.
Armstrong in describing his affinities is certainly dealing with quantised units, of six for benzene for example. He is describing altered distributions of these integer objects. Three years earlier in 1887, he had published two tables, one describing those radicles that induced o/p substitutions and another inducing m-substitutions. He was therefore developing his ideas about what the differences between these two classes might be. So when he describes the altered distribution of an affinity brought about by a radicle as then having an effect that induces positional differences in reactivity, he is not that very far from how we think about it nowadays.Try replacing his word affinity by the modern electron to see if his statements continue to make sense.
I understand the point. However in my opinion every “translation” needs a “background”. There are many points in this translation that we assume “for granted” and obvious facts which is not the case. The background atomic and structural theory which are obvious to us have not been recognized in detail in late 19 century. Even in 1905 there were yet serious opponents to the atomic theory. Even practitioners of the Structural theory was quite cautioned to attribute “reality” to molecular structures (I have insisted on this point in one of my previous posts on Kekule’s oscillation theory of Benzene). So, while it seems to me that it is wise to claim that some theories of 19 century organic chemistry were innovative and grasp the reality to some extent but certainly they are not comparable to modern post-quantum mechanics era. I am inclined to accept Thomas Kuhn approach (described in “Structure of Scientific Revolutions”) in considering the history of science. By the way, I understand this is a difference in opinion.
It is of course difficult to speculate about what was in Armstrong’s mind when he wrote those words (one would probably need to track down his letters and correspondence to other scientists to get some insights). I think it useful to say that anyone reading those words nowadays could very easily say that they recognise the modern interpretation in the prose, and even the problem he is trying to solve (he speculated about what the differences might be that make one radicle o/p direct and another m-direct. That is a very modern question to ask). One could for example nowadays show this to a student tutorial group without worries, and expect them to understand the issues quite quickly.
I did try to read some of Robinson’s articles criticising Hückel you kindly provided DOIs for. These are NOT readable nowadays and I would not show them to modern students. Robinson was also famously combative (did I detect quite some antipathy to Lewis in his 1922 article?). The articles by Hückel himself are also regarded by many as unreadable.
Curiously however, when I was trying to understand Kekule’s famous 1872 paper on the oscilation/vibration in benzene, it was Armstrong who quickly provided a translation (also in 1872); doi: 10.1039/JS8722500605 and which in turn every student of organic chemistry with poor German could be shown in a tutorial nowadays. They should also view another 1872 interpretation of Kekule’s words, this time as a picture doi:10.1002/cber.187200501139. We may again argue whether writing in 1872, any of these authors could possibly have understood the problem in anything like the manner we do nowadays, but I contend that at least a modern chemistry student would have little difficulty in immediately grasping the ideas and how they have evolved.