Ten years are a long time when it comes to (recent) technologies. The first post on this blog was on the topic of how to present chemistry with three intact dimensions. I had in mind molecular models, molecular isosurfaces and molecular vibrations (arguably a further dimension). Here I reflect on how ten years of progress in technology has required changes and the challenge of how any necessary changes might be kept “under the hood” of this blog.
Posts Tagged ‘jmol’
Twenty one years of chemistry-related Java apps: RIP Java?
Saturday, June 10th, 2017In an earlier post, I lamented the modern difficulties in running old instances of Jmol, an example of an application program written in the Java programming language. When I wrote that, I had quite forgotten a treasure trove of links to old Java that I had collected in 1996-7 and then abandoned. Here I browse through a few of the things I found.
Does combining molecules with augmented reality have a future?
Monday, March 28th, 2016Augmented reality, a superset if you like of virtual reality (VR), has really been hitting the headlines recently. Like 3D TV, its been a long time coming! Since ~1994 or earlier, there have been explorations of how molecular models can be transferred from actual reality to virtual reality using conventional computers (as opposed to highly specialised ones). It was around then that a combination of software (Rasmol) and hardware (Silicon Graphics, and then soon after standard personal computers with standard graphics cards) became capable of such manipulations. VRML (virtual reality modelling language) also proved something of a false start‡ So have things changed?
WebCite and Jmol
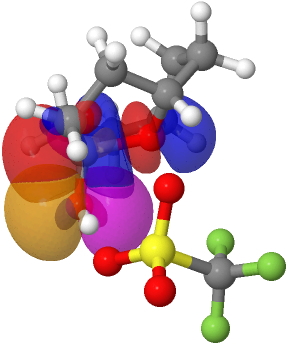
Sunday, April 18th, 2010A Disrotatory 4n+2 electron anti-aromatic Möbius transition state for a thermal electrocyclic reaction.
Thursday, April 2nd, 2009Mauksch and Tsogoeva have recently published an article illustrating how a thermal electrocyclic reaction can proceed with distoratory ring closure, whilst simultaneously also exhibiting 4n electron Möbius-aromatic character[1]. Why is this remarkable? Because the simple Woodward-Hoffmann rules state that a disrotatory thermal electrocyclic reaction should proceed via a Hückel-aromatic 4n+2 electron transition state. Famously, Woodward and Hoffmann stated there were no exceptions to this rule. Yet here we apparently have one! So what is the more fundamental? The disrotatory character, or the 4n/Möbius character in the example above? Mauksch and Tsogoeva are in no doubt; it is the former that gives, and the latter is correct.
References
- M. Mauksch, and S. Tsogoeva, "A Preferred Disrotatory 4<i>n</i> Electron Möbius Aromatic Transition State for a Thermal Electrocyclic Reaction", Angewandte Chemie International Edition, vol. 48, pp. 2959-2963, 2009. https://doi.org/10.1002/anie.200806009