2. Historical Background.
Prior to the 1960s, organic reactivity was thought to be dominated by
factors such as
- the relative stability of reactant, transition state and product (ie thermodynamics),
- geometrical effects such as strain and steric interactions, hydrogen bonding,
- electrostatic effects such as the polarity of functional groups (eg the carbonyl group) and
- the aromaticity of either the reactant or the product.
During the course of the synthesis of vitamin B12 in the early 1960s, R. B. Woodward noticed that
none of the above factors could rationalise the following reaction, since the actual product is
sterically the LEAST stable.
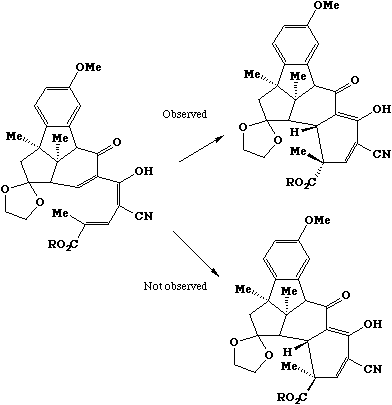
A new explanation was proposed based on 'stereoelectronic' factors, ie recognising that the three-
dimensional properties of the electrons and their phase relationship could dominate the other factors
listed above. This theory became known as 'the conservation of orbital symmetry', and together with
the theoretician Roald Hoffmann, Woodward was eventually awarded the Nobel prize for this work.
This recognition that the 'shape' of the electrons surrounding a molecule, or more accurately the three
dimensional properties of the 'molecular wavefunction' was critical to explaining reactivity has had
an enormous impact on understanding organic chemistry.
Return to index

