White City is a small area in west london created as an exhibition site in 1908, morphing over the years into an Olympic games venue, a greyhound track, the home nearby of the BBC (British Broadcasting Corporation) and most recently the new western campus for Imperial College London.♣ The first Imperial department to move into the MSRH (Molecular Sciences Research Hub) building is chemistry. As a personal celebration of this occasion, I here dedicate three transition states located during my first week of occupancy there, naming them the White City trio following earlier inspiration by a string trio and their own instruments.
Posts Tagged ‘acetic acid’
The mechanism of ester hydrolysis via alkyl oxygen cleavage under a quantum microscope
Tuesday, April 2nd, 2013My previous dissection of the mechanism for ester hydrolysis dealt with the acyl-oxygen cleavage route (red bond). There is a much rarer[1] alternative: alkyl-oxygen cleavage (green bond) which I now place under the microscope.
References
- C.A. Bunton, and J.L. Wood, "Tracer studies on ester hydrolysis. Part II. The acid hydrolysis of tert.-butyl acetate", Journal of the Chemical Society (Resumed), pp. 1522, 1955. https://doi.org/10.1039/jr9550001522
The mechanism (in 4D) of the reaction between thionyl chloride and a carboxylic acid.
Friday, May 25th, 2012If you have not previously visited, take a look at Nick Greeves’ ChemTube3D , an ever-expanding gallery of reactions and their mechanisms. The 3D is because all molecules are offered with X, Y and z coordinates. You also get arrow pushing‡ in 3D. Here, I argue that we should adopt Einstein, and go to the space-time continuum! By this, I mean one must also include the order in which things happen. To my knowledge, no compendium of (organic) reaction mechanisms incorporates this 4th dimension. My prelude to this post nicely illustrated this latter aspect. Here I continue with an exploration of the mechanism of forming an acyl chloride from a carboxylic acid using thionyl chloride. The mechanism shown at ChemTube3D is as below and will now be tested for its reasonableness using quantum mechanics.
Surprises (?) in the addition of HCl to a carbonyl group.
Thursday, May 24th, 2012HCl reacting with a carbonyl compound (say formaldehyde) sounds pretty simple. But often the simpler a thing looks, the more subtle it is under the skin. And this little reaction is actually my prelude to the next post.
The oldest reaction mechanism: updated!
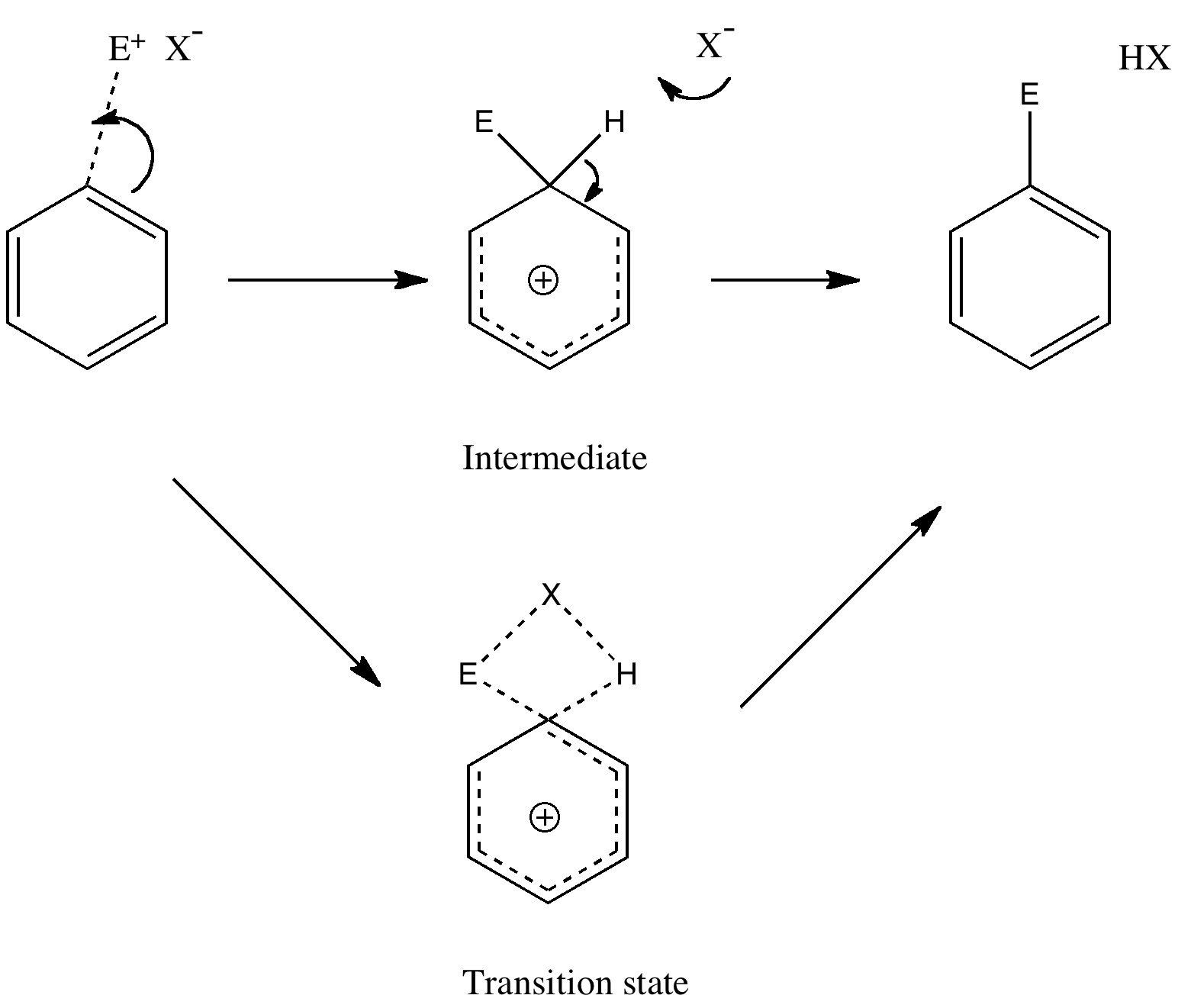
Tuesday, September 14th, 2010Unravelling reaction mechanisms is thought to be a 20th century phenomenon, coincident more or less with the development of electronic theories of chemistry. Hence electronic arrow pushing as a term. But here I argue that the true origin of this immensely powerful technique in chemistry goes back to the 19th century. In 1890, Henry Armstrong proposed what amounts to close to the modern mechanism for the process we now know as aromatic electrophilic substitution [1]. Beyond doubt, he invented what is now known as the Wheland Intermediate (about 50 years before Wheland wrote about it, and hence I argue here it should really be called the Armstrong/Wheland intermediate). This is illustrated (in modern style) along the top row of the diagram.
References
- "Proceedings of the Chemical Society, Vol. 6, No. 85", Proceedings of the Chemical Society (London), vol. 6, pp. 95, 1890. https://doi.org/10.1039/pl8900600095