I previously blogged about anomeric effects involving π electrons as donors, and my post on the conformation of 1,2-difluorethane turned out one of the most popular. Here I thought I would present the results of searching the Cambridge crystal database for examples of the gauche effect. The basic search is defined below
Here, we define a four-atom torsion (TOR1), the two central carbon atoms having two groups R which can be only H or C. These two carbons are also defined as acyclic. The restrictions of the search as defined above also include R-factor < 0.05, not disordered and no errors. These combine to reduce the number of hits significantly (although not dissimilar distributions are obtained for less restricted searches). Each search takes only a few seconds, and one can rattle through many permutations very quickly.
So here come the results. First, QA=4M=F. All but one of the examples has a torsion in the region of 60°, the classic gauche effect!
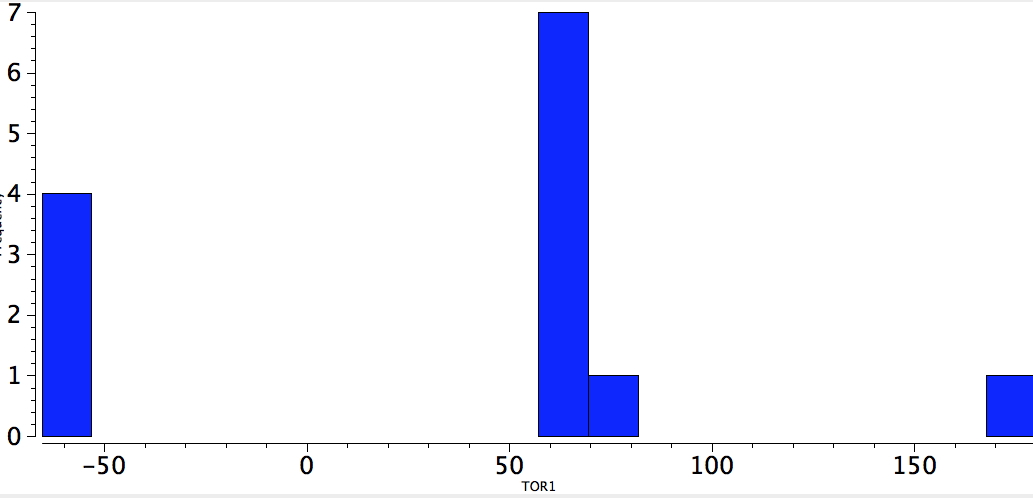
F-C-C-F
Next, QA=O, 4M=F. Rather more hits, and the effect is almost as clear-cut. I should point out that the apparent "exceptions" to the gauche conformation may arise from structural restrictions, and each really would have to be inspected individually for the reasons (which I do not attempt here).
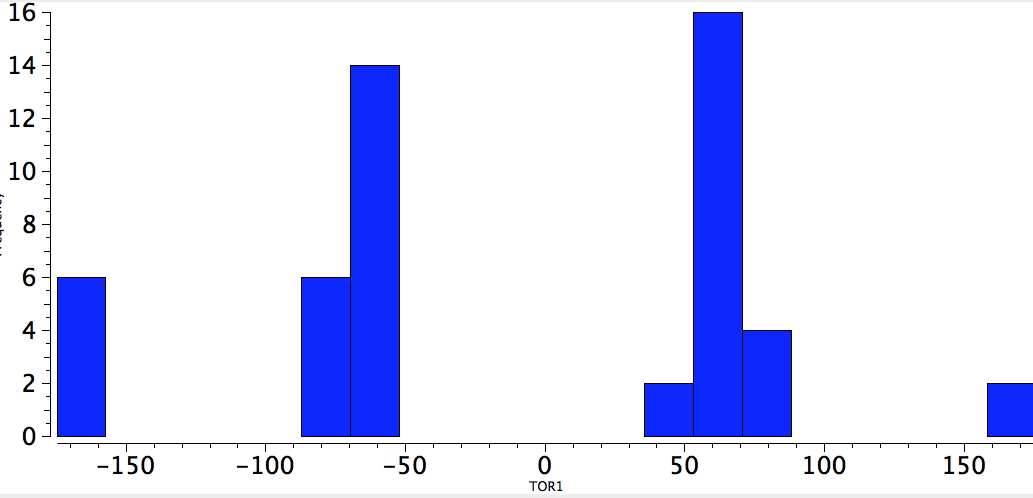
OCCF
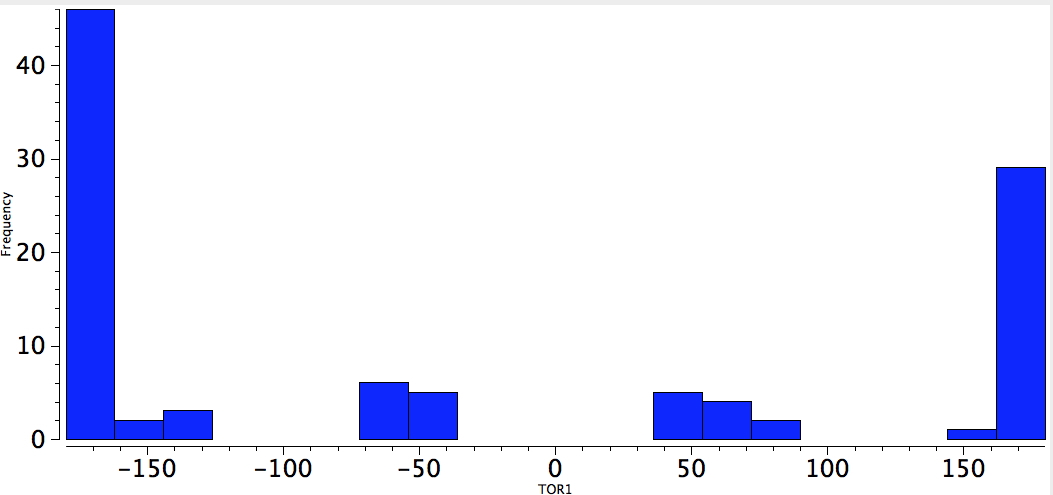
With QA=4M=O, one has many more instances. The effect is pretty convincing (it may be that hydrogen bonding may also control the conformation).
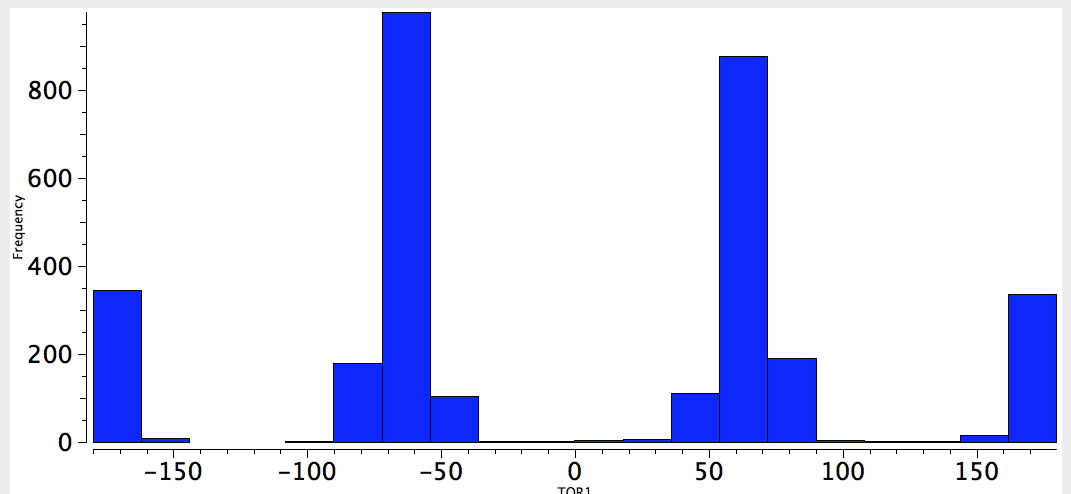
O-C-C-O
Now for QA=4M=Cl. The distribution is slanted more to the anti conformation, but there are still quite a few gauche.
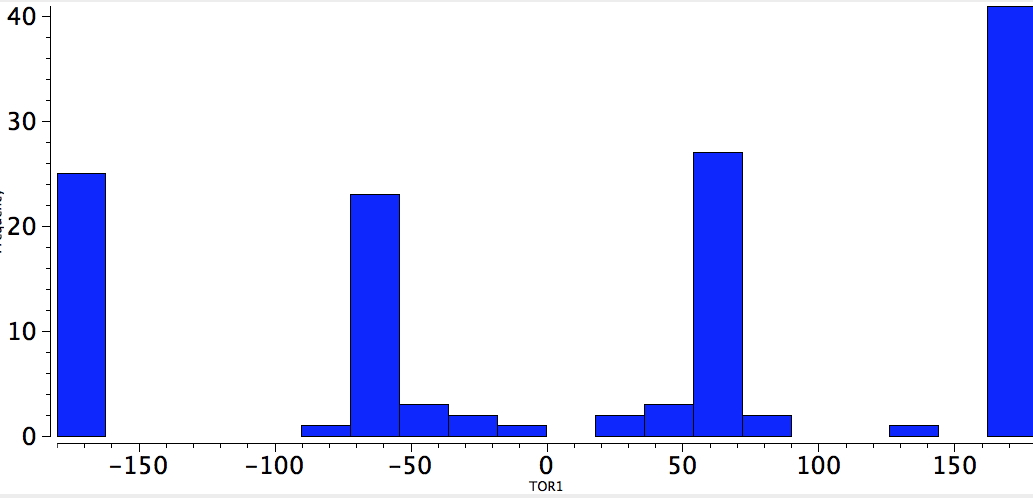
Cl-CC-Cl
With QA=4M=S, the conformations are now almost all anti; the gauche effect is no more!
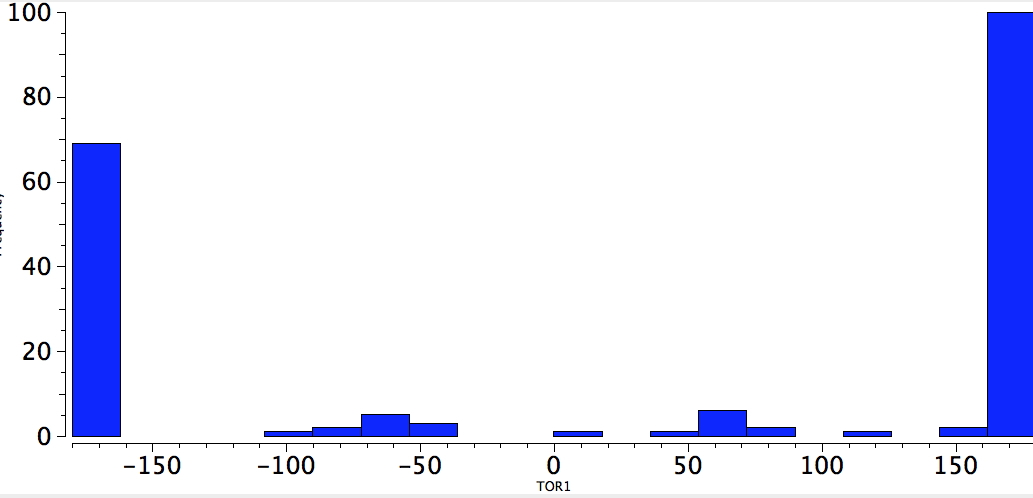
S-C-C-S
And for QA=4M=Br, it has also almost vanished (there is only one instance for I, and that too is antiperiplanar).
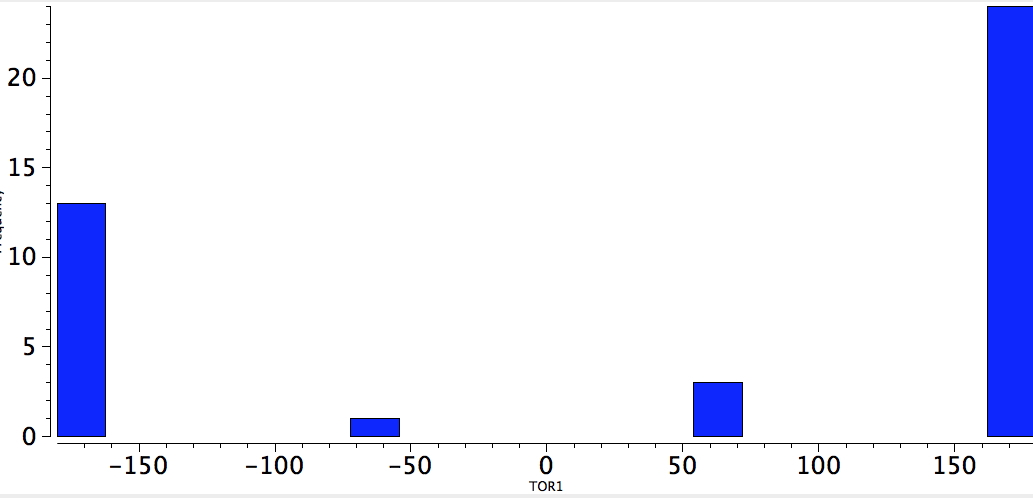
Br-C-C-Br
I now return to an earlier post in which I speculated that a cyano group might participate in the anomeric effect. Well here it is in the gauche effect; QA=CN, 4M = any of N,O,F,Cl,S. Quite a few gauche orientations for this pseudo-halogen!
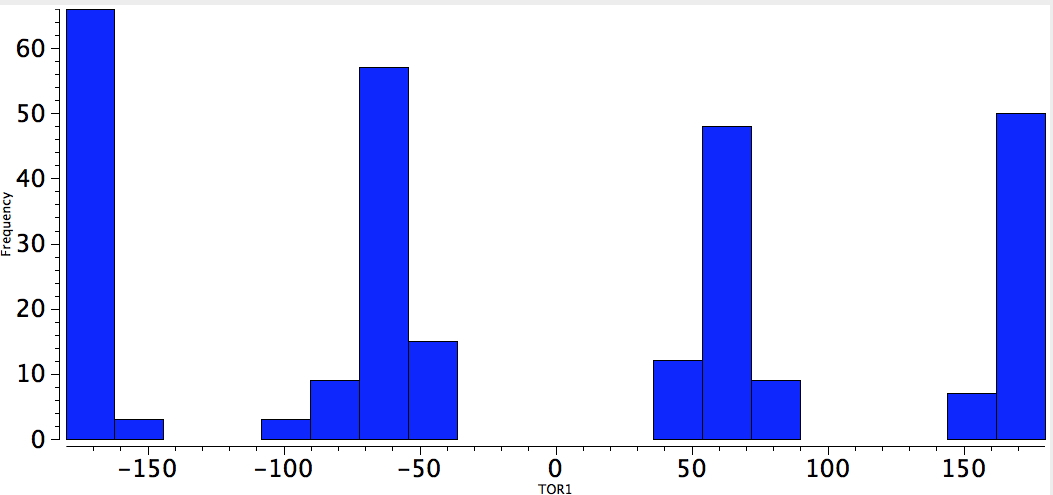
Neg-C-C-CN
Another group that can act as a powerful acceptor of electrons from a donor is QA=N(Me)3+.. With 4M= N, O, F, Cl, here the population of gauche conformers is large. QA=CF3 is a similar group.
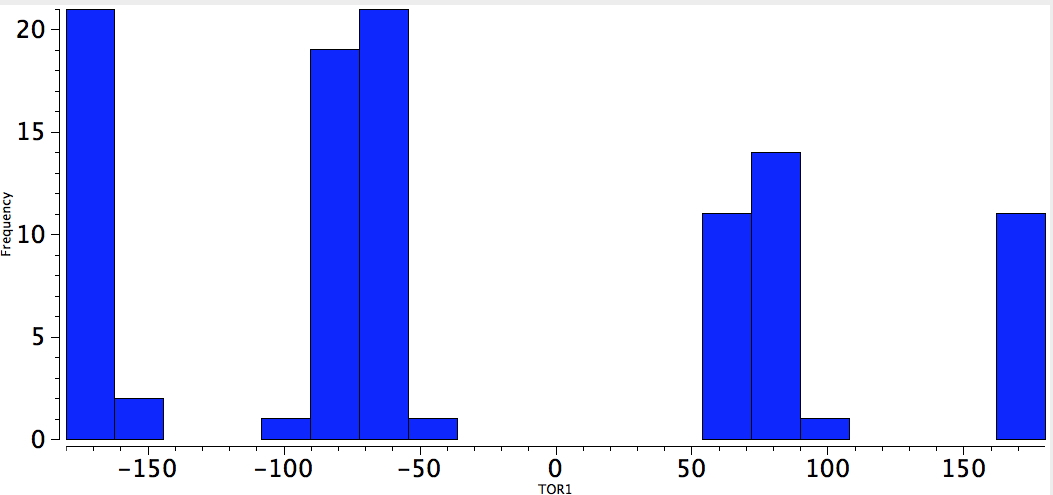
Neg-C-C-NMe3
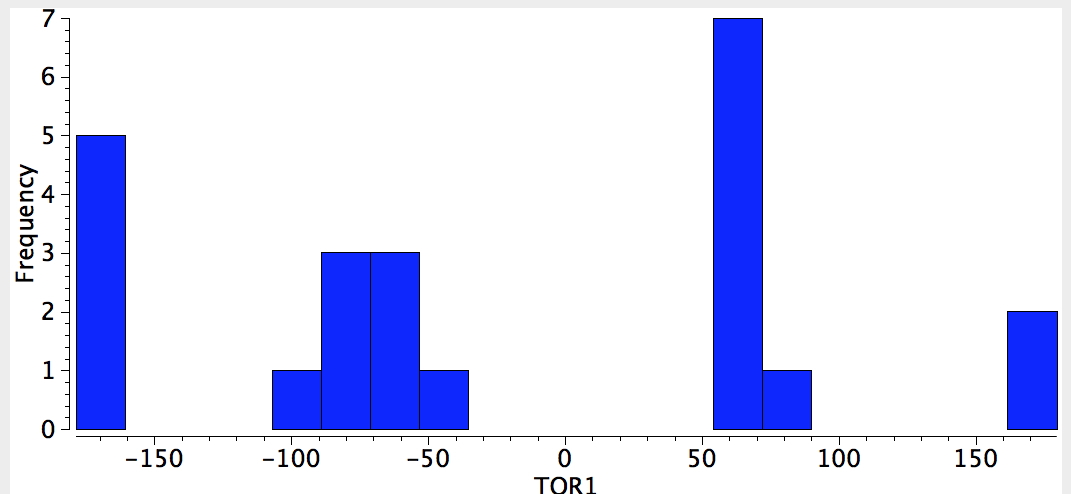
Neg-C-C-CF3
One can envisage other combinations. Thus QA= C=C, 4M = any of N, O, F, Cl. An alkene seems one of the more powerful gauche effect participants!
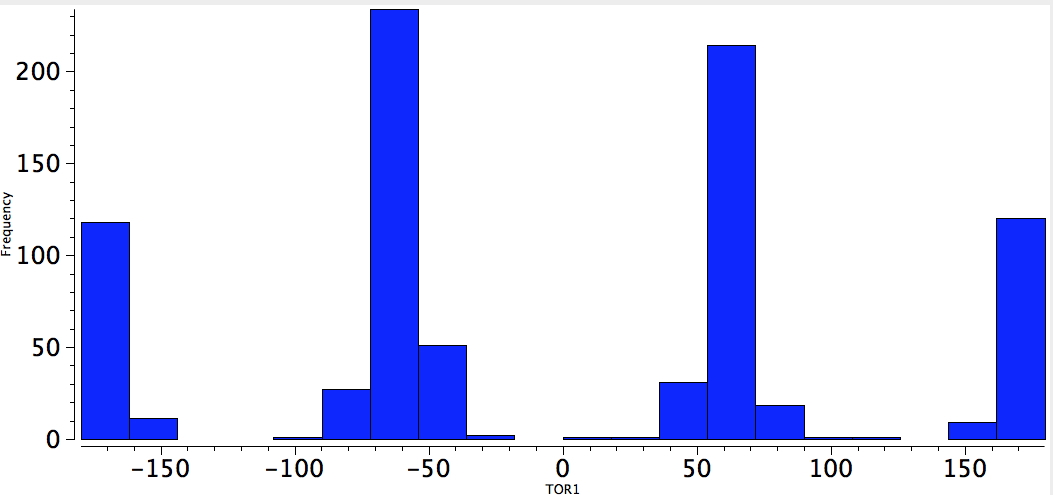
alkene-C-C-Neg
And alkynes, perhaps slightly less so.
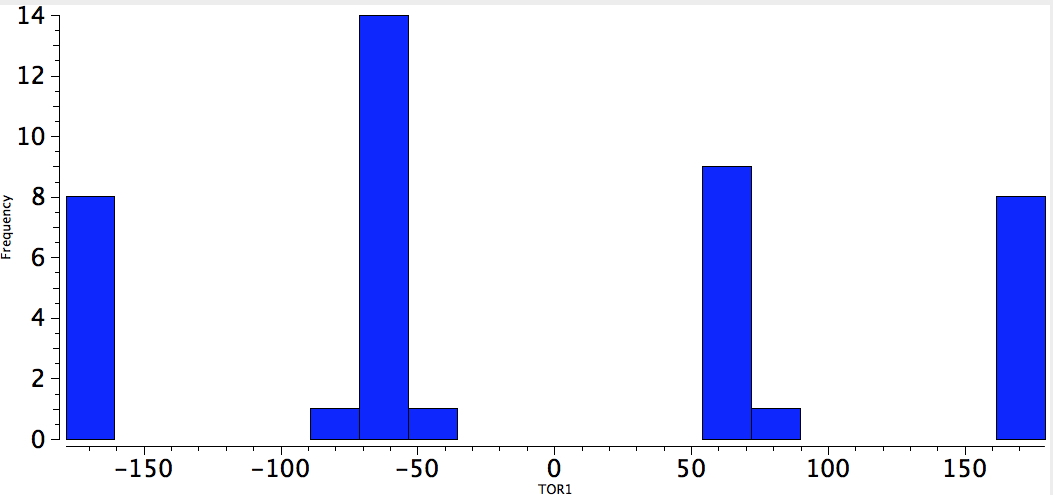
Alkyne-C-C-Neg
What about metals (QA = any metal, 4M = any of N, O, F, Cl, S). Well, not particularly biased either way, but clearly one in which the identity of the metal may matter.
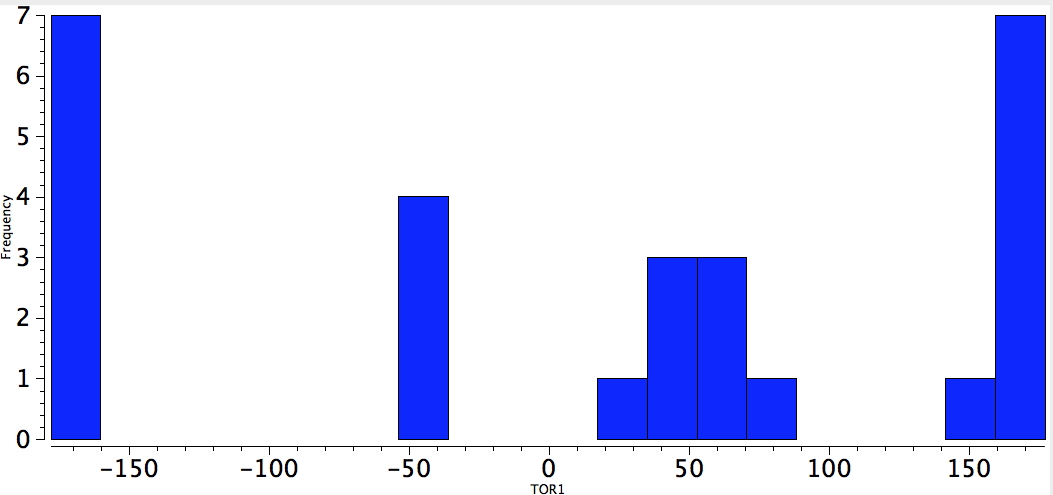
Metal-C-C-electronegative
I should end with inverting the model. If QA is electropositive (any group to the left of carbon, or below it in the periodic table) and 4M is electronegative, than they align almost exclusively anti-periplanar and not gauche. But notice how relatively few examples there are. Synthetic chemists, please make more such molecules!
Electropositive-C-C-Electronegative
If you thought the gauche effect was restricted to just a few molecules, think again!
Acknowledgments
This post has been cross-posted in PDF format at Authorea.
Tags: basic search, Cambridge, CF 3, conformational analysis, gauche, Interesting chemistry, metal, search takes, similar, Tutorial material
These results illustrate the effectiveness of such quick search methods for providing preliminary information, and point to the need for a thorough computational study. One would like to have a table of gauche-anti energy differences in simple XCH2CH2Y systems as well as an analysis of the effects responsible.
I agree totally. This type of survey is more of a tease than a thorough analysis of any individual system.
I would point out that the Cambridge database, with some 600,000 entries, is probably a very evenly distributed system, which by this virtue carries lots of unintended effects not anticipated by the people who made the molecules. All we have to do is discover them.
I might also add that it is estimated that the total number of crystal structures done to date is probably four times larger; only about 25% of structures solved are actually deposited there (the rest languish in the unpublished drawer). If we did have the full database (let us say 2.5 million entries) then the chances of such unexpected discoveries really do go up.
A very interesting survey, thank you! I wish I have thought about detecting gauche effect this way when I was writing my PhD thesis.
However, it mostly discussed a new orbital-free way to analyze the generalized anomeric effect (“lone pair – X- C – Y”) using QTAIM and the gauche effect might not fit in.
I must admit, my PhD thesis also included a separate subchapter concerning the anomeric effect involving the CN groups, which has yet to be published (the effect is generally really small). As for more exotic cases, I have found that the cyclopropyl group can be a donor in an anomeric interaction (much like the case for alkenes you have described elsewhere in this blog).
If you are interested, I can provide some references, but I do not want to appear as a spammer here 🙂
The more references the better!
Regarding the anomeric effect involving CN groups, did you see this post? For that particular structure, it seems reasonably clear-cut?
I have also done a quick search of the CCDC for alignments between one C-C bond in cyclopropyl (as a donor) and the C-O of an adjacent C-O bond (as an acceptor). The search was done by restricting the C-C bond connecting the C-O and the C-C of the cyclopropyl to be acyclic. The anomeric effect can operate at angles of ~150-180° and the clustering around this value below seems reasonably prominent.

This plot is for C-X (X=F, Cl) as the acceptor (with the restriction that two other substituents on the carbon bearing the halogen must be R=H, C, i.e. not another halogen) shows not many examples, but perhaps again a bias towards an anomeric interaction?
My own example considered changes in C-Cl bond lengths (doi:10.1007/s11172-009-0131-2), and the methodology used is described in detail here: doi:0.1021/jp800384k1. The O-C-CN interaction described in the post by your link seems completely legit, but I have some doubts about the importance of the C=C-C-CN one. It should also be noted that in my experience the CN group is the only case of a reasonably strong anomeric interaction with C atom as an acceptor: CF3 and even C(NO2)3 do not seem to work.
I guess that the gauche effect is very sensitive to the interaction between the QA and 4M groups. Namely, the absence of S-S and Br-Br “gauches” is most likely caused by the “hockey sticks” effect (doi:10.1021/jp203730b).
[…] subtle in neutral molecules, but following my use of crystal structures to explore the so-called gauche effect (which originates from σ-σ-conjugation), I thought I would have a go here at seeing what the […]
Dear Henry, I hope to see this analysis published, so I can include it in my book!
Igor,
Ah, one of many items on the list!